- About
- Browse Articles
-
Special Issues
- Pioneering strategies for overcoming bacterial drug resistance (2026)
- Advancing microbial engineering through synthetic biology (2025)
- Host-associated microbiome (2024)
- Bacterial regulatory mechanisms for the control of complex cellular mechanisms (2023)
- Two years into COVID-19 pandemic: Where are we? (2022)
- Collections
- For Contributors
- Policies
- E-Submission

- About
- Browse Articles
-
Special Issues
- Pioneering strategies for overcoming bacterial drug resistance (2026)
- Advancing microbial engineering through synthetic biology (2025)
- Host-associated microbiome (2024)
- Bacterial regulatory mechanisms for the control of complex cellular mechanisms (2023)
- Two years into COVID-19 pandemic: Where are we? (2022)
- Collections
- Policies
- For Contributors
Articles
- Page Path
- HOME > J. Microbiol > Volume 64(3); 2026 > Review
-
Review
Antibiotic hybrids: A promising strategy to replenish the pipeline and combat antimicrobial resistance - Yeongseo Lee1,2, Yeo Jin Kim1,3, Minhee Oh1,3, Joon-Ho Lee1,4,*, Saemee Song1,*, Jaesung Kwak1,2,*
-
Journal of Microbiology 2026;64(3):e2510006.
DOI: https://doi.org/10.71150/jm.2510006
Published online: February 25, 2026
1Infectious Diseases Therapeutic Research Center, Korea Research Institute of Chemical Technology (KRICT), Daejeon 34114, Republic of Korea
2Division of Medicinal Chemistry and Pharmacology, KRICT School, University of Science and Technology (UST), Daejeon 34114, Republic of Korea
3School of Pharmacy, Sungkyunkwan University, Suwon 16419, Republic of Korea
4Graduate School of New Drug Discovery and Development, Chungnam National University, Daejeon 34134, Republic of Korea
- *Correspondence Joon-Ho Lee jh_k@krict.re.kr Saemee Song saemee@krict.re.kr Jaesung Kwak jkwak@krict.re.kr
© The Microbiological Society of Korea
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Antimicrobial resistance (AMR) poses an ongoing threat to global health, with the number of deaths directly attributable to AMR projected to rise to 8 million. One of the main reasons for the current crisis is the depletion of antibiotic candidates in clinical pipelines. To address this, more preclinical candidates must be advanced into development. However, the scientific challenges and limited economic incentives associated with antibiotic research have further aggravated the situation. Antibiotic hybrids, which combine two antibiotics with different modes of action, have emerged as a promising strategy to overcome AMR and are already being developed for clinical use. This approach takes advantage of the strong selective pressure exerted when two bactericidal agents act simultaneously. Importantly, because hybrids are administered as a single chemical entity, they may offer advantages over conventional combination therapies, such as simplified pharmacokinetics and dosing. Furthermore, since clinically validated antibiotics are used as the building blocks of hybrids, this strategy provides an efficient platform for generating new lead compounds. Recently, the concept of antibiotic hybrids has expanded beyond antibiotic–antibiotic conjugates to include the attachment of functional molecules designed to mitigate the disadvantages of the parent antibiotics. In this review, we summarize the definition of antibiotic hybrids, highlight representative compounds that have entered clinical evaluation, and discuss recent advances in their development.
Introduction
Challenges and Strategies of Antibacterial Drug Discovery
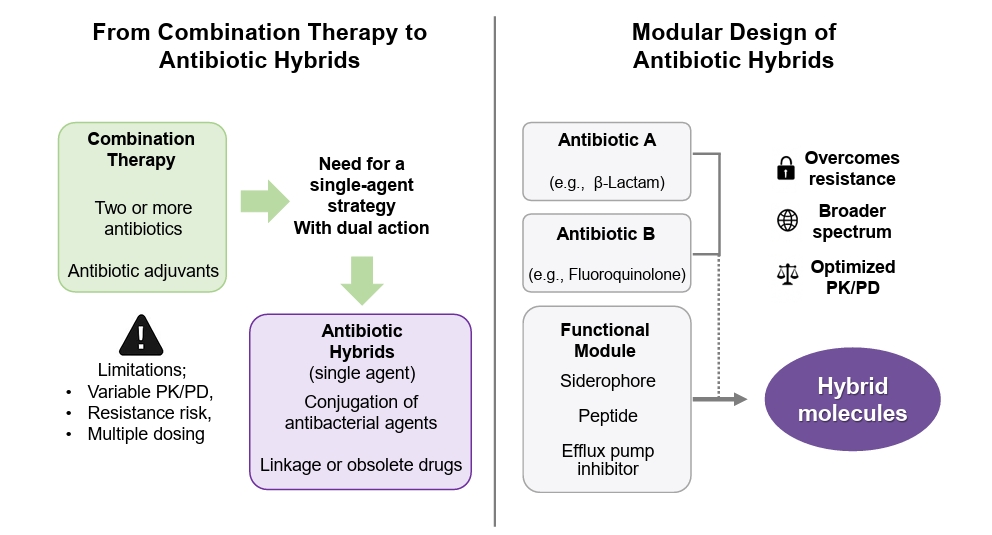
Combination Therapy
Antibiotic Hybrid Strategy
Recent Development of Antibiotic Hybrids (2021–2025)
Antibiotic Hybrids in Clinical Trials
Future Outlooks
Acknowledgments
This work was supported by the R&D program of the Institutional Research Program of the KRICT (KK2633-30).
Conflict of Interest
The authors declare no competing financial interest.
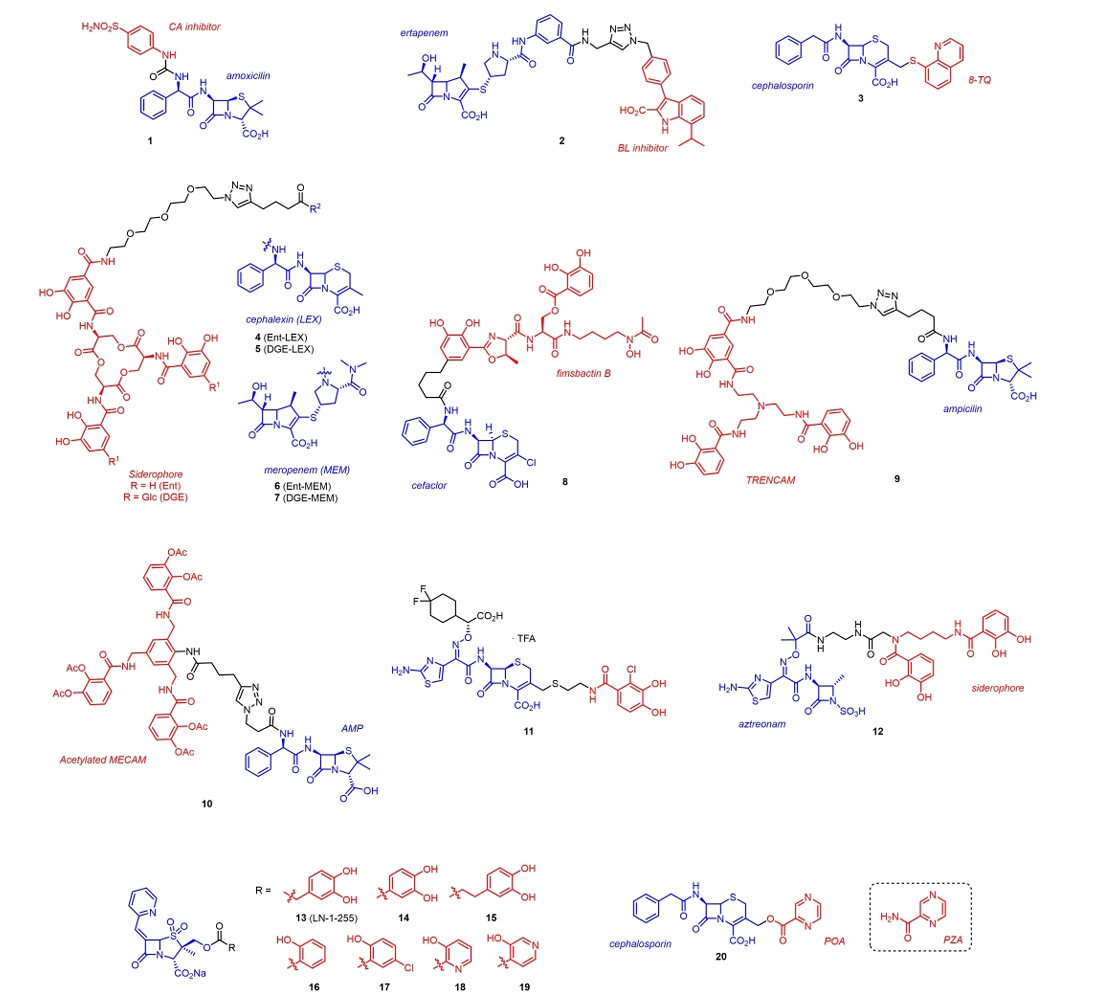
| Cpd. | Hybrid design | Target | Efficacy | Reference |
|---|---|---|---|---|
| 1 | Amoxicillin + CA inhibitor | N. gonorrhoeae (CDC-178) | MIC, 1 (1) vs 2 (amoxicillin) μg/ml | Bonardi et al. (2024) |
| 2 | Carbapenem + MBL Inhibitor | MBL Enzymes (NDM-1, IMP-1) | High Enzyme Inhibition (IC50 nM range), Inactive in cells | Gao et al. (2024) |
| 3 | Cephalosporin + 8-TQ | K. pneumoniae (MBL-producer) | Restored efficacy of Meropenem via enzyme-triggered release of 8-TQ. | van Haren et al. (2021) |
| 6 | Meropenem + Enterobactin | E. coli (CFT073) | MIC, 0.01 (6) vs 0.1 (Meropenem) μM | Sargun et al. (2021) |
| 8 | Cefaclor + Fimsbactin B | A. baumannii (ATCC 17978) | MIC, 0.5 (8) vs 64 (Cefaclor) μg/ml | Kim and Kim (2021) |
| 9 | Ampicillin + TRENCAM | S. Typhimurium | MIC, 0.01 (9) vs 10 (Ampicillin) μM | Motz et al. (2024) |
| 10 | Ampicillin + MECAM | E. coli | MIC, 1.5 (10) vs 46 (Ampicillin) μM | Pinkert et al. (2021) |
| 11 | Cephalosporin + Catechol | A. baumannii (MDR Isolate) | MIC, 0.25 (11) vs ≥ 64 (Meropenem) μg/ml | Liu et al. (2024) |
| 20 | Cephalosporin + POA | M. tuberculosis (H37Rv ΔpncA) | MIC, 100 (20) vs > 800 (PZA) vs 100 (POA) μg/ml | Cole et al. (2022) |
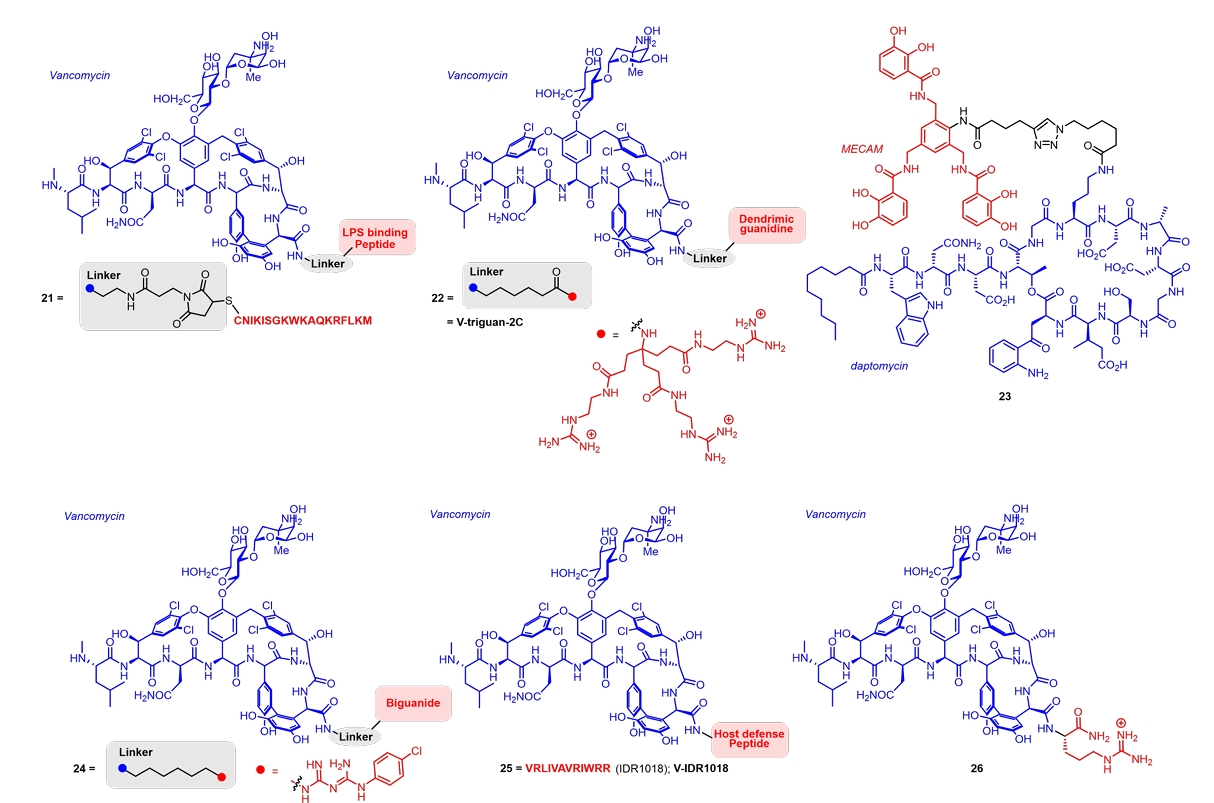
| Cpd. | Hybrid design | Target | Efficacy | Reference |
|---|---|---|---|---|
| 21 | Vancomycin + LPS-binding peptide | E. coli (AB1157) | MIC, 8 (21) vs > 88 (Vancomycin) μM | Shi et al. (2021) |
| 22 | Vancomycin + Dendrimeric guanidine | E. coli (UTI89) | MIC, 8 (22) vs 128 (Vancomycin) µM | Chosy et al. (2024) |
| 23 | Daptomycin + MECAM | A. baumannii | MIC, 4.4 (23) vs > 39 (Daptomycin) µM | Pinkert et al. (2021) |
| 24 | Vancomycin + Biguanide | E. faecium (ATCC 51559) | MIC, 4 (24) vs 512 (Vancomycin) µM | Rahn et al. (2024) |
| 25 | Vancomycin + IDR1018 peptide | MRSA biofilms | Minimal biofilm eradication conc. 16 (25) vs > 128 (Vancomycin) µg/ml | Etayash et al. (2021) |
| 26 | Vancomycin + Arginine | M. abscessus (ATCC 19977) | MIC, 16 (26) vs 64 (Vancomycin) µM | Brčić et al. (2023) |
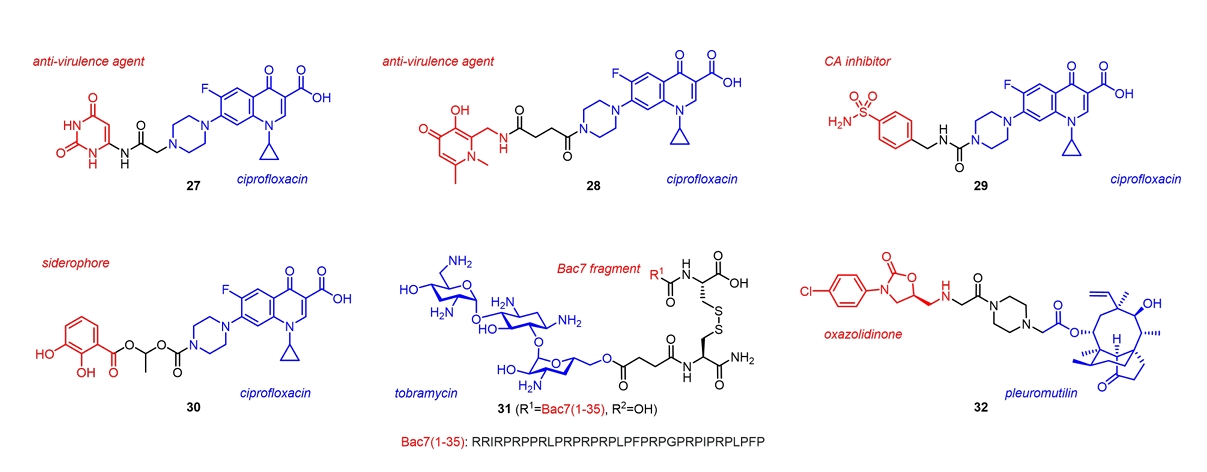
| 27 | Ciprofloxacin + Uracil | MRSA (AUMC261) | MIC, 0.031 (27) vs 0.57 (Ciprofloxacin) µM | Samir et al. (2022) |
| 28 | Ciprofloxacin + Anti-virulence agent | P. aeruginosa Biofilms | Biofilm reduction at 1/4 MIC, 78.3% (28) vs 12.6% (Ciprofloxacin) | Wang et al. (2023) |
| 30 | Ciprofloxacin + Siderophore | B. pseudomallei (BPS2020IRBA003) | MIC, 2 (30) vs 16 (Ciprofloxacin) µg/ml | Loupias et al. (2023) |
| 31 | Tobramycin + Bac7 peptide fragment | E. coli (BW25113) | MIC, 1 (31) vs 4 (Tobramycin) µM | Gambato et al. (2023) |
| 32 | Pleuromutilin + Oxazolidinone | MRSA (ATCC33591) | MIC, 0.063 (32) vs > 0.5 (Pleuromutilin) μg/ml | Xia et al. (2023) |
- Ackley R, Roshdy D, Meredith J, Minor S, Anderson WE, et al. 2020. Meropenem-vaborbactam versus ceftazidime-avibactam for treatment of carbapenem-resistant Enterobacteriaceae infections. Antimicrob Agents Chemother. 64: e02313-19–19. ArticlePubMedPMCLink
- Alarcia-Lacalle A, Canut-Blasco A, Solinís MA, Isla A, Rodríguez-Gascón A. 2025. Clinical efficacy, safety and pharmacokinetics of novel β-lactam/β-lactamase inhibitor combinations: a systematic review. JAC Antimicrob Resist. 7: dlaf096.ArticlePubMedPMCPDF
- Albrecht HA, Beskid G, Chan KK, Christenson JG, Cleeland R, et al. 1990. Cephalosporin 3′-quinolone esters with a dual mode of action. J Med Chem. 33: 77–86. ArticlePubMed
- Baldoni D, Gutierrez M, Timmer W, Dingemanse J. 2014. Cadazolid, a novel antibiotic with potent activity against Clostridium difficile: safety, tolerability and pharmacokinetics in healthy subjects following single and multiple oral doses. J Antimicrob Chemother. 69: 706–714. ArticlePubMed
- Belachew WA, Naafs B. 2019. Position statement: LEPROSY: diagnosis, treatment and follow-up. J Eur Acad Dermatol Venereol. 33: 1205–1213. ArticlePubMed
- Bergkessel M, Forte B, Gilbert IH. 2023. Small-molecule antibiotic drug development: need and challenges. ACS Infect Dis. 9: 2062–2071. ArticlePubMedPMCLink
- Beskid G, Siebelist JA, McGarry CM, Cleeland R, Chan K, et al. 1990. In vivo evaluation of a dual-action antibacterial, Ro 23-9424, compared to cefotaxime and fleroxacin. Chemotherapy. 36: 109–116. ArticlePubMedLink
- Bilal M, El Tabei L, Busker S, Krauss C, Fuhr U, et al. 2021. Clinical pharmacokinetics and pharmacodynamics of cefiderocol. Clin Pharmacokinet. 60: 1495–1508. ArticlePubMedPMCPDF
- Blais J, Lewis SR, Krause KM, Benton BM. 2012. Antistaphylococcal activity of TD-1792, a multivalent glycopeptide-cephalosporin antibiotic. Antimicrob Agents Chemother. 56: 1584–1587. ArticlePubMedPMCLink
- Boattini M, Bianco G, Charrier L, Comini S, Iannaccone M, et al. 2023. Rapid diagnostics and ceftazidime/avibactam for KPC-producing Klebsiella pneumoniae bloodstream infections: impact on mortality and role of combination therapy. Eur J Clin Microbiol Infect Dis. 42: 431–439. ArticlePubMedPDF
- Bonardi A, Nocentini A, Giovannuzzi S, Paoletti N, Ammara A, et al. 2024. Development of penicillin-based carbonic anhydrase inhibitors targeting multidrug-resistant Neisseria gonorrhoeae. J Med Chem. 67: 9613–9627. ArticlePubMedPMC
- Brčić J, Tong A, Wender PA, Cegelski L. 2023. Conjugation of vancomycin with a single arginine improves efficacy against mycobacteria by more effective peptidoglycan targeting. J Med Chem. 66: 10226–10237. ArticlePubMedPMCLink
- Bryskier A. 1997. Dual β-lactam-fluoroquinolone compounds: a novel approach to antibacterial treatment. Expert Opin Invest Drugs. 6: 1479–1499. Article
- Carmeli Y, Cisneros JM, Paul M, Daikos GL, Wang M, et al. 2025. Aztreonam-avibactam versus meropenem for the treatment of serious infections caused by Gram-negative bacteria (REVISIT): a descriptive, multinational, open-label, phase 3, randomised trial. Lancet Infect Dis. 25: 218–230. ArticlePubMed
- Centers for Disease Control and Prevention. 2024. Antimicrobial resistance threats in the United States; 2021–2022. Available from https://www.cdc.gov/antimicrobial-resistance/data-research/threats/update-2022.html
- Chan BC, Ip M, Lau CB, Lui SL, Jolivalt C, et al. 2011. Synergistic effects of baicalein with ciprofloxacin against NorA over-expressed methicillin-resistant Staphylococcus aureus (MRSA) and inhibition of MRSA pyruvate kinase. J Ethnopharmacol. 137: 767–773. ArticlePubMed
- Chosy MB, Sun J, Rahn HP, Liu X, Brčić J, et al. 2024. Vancomycin-polyguanidino dendrimer conjugates inhibit growth of antibiotic-resistant Gram-positive and Gram-negative bacteria and eradicate biofilm-associated Staphylococcus aureus. ACS Infect Dis. 10: 384–397. ArticlePubMedPMCLink
- Christenson JG, Chan KK, Cleeland R, Dix-Holzknecht B, Farrish HH Jr, et al. 1990. Pharmacokinetics of Ro 23-9424, a dual-action cephalosporin, in animals. Antimicrob Agents Chemother. 34: 1895–1900. ArticlePubMedPMCLink
- Cole MS, Howe MD, Buonomo JA, Sharma S, Lamont EA, et al. 2022. Cephem-pyrazinoic acid conjugates: circumventing resistance in Mycobacterium tuberculosis. Chem Eur J. 28: e202200995. ArticlePubMedLink
- Cornely OA, Cisneros JM, Torre-Cisneros J, Rodríguez-Hernández MJ, Tallón-Aguilar L, et al. 2020. Pharmacokinetics and safety of aztreonam/avibactam for the treatment of complicated intra-abdominal infections in hospitalized adults: results from the REJUVENATE study. J Antimicrob Chemother. 75: 618–627. ArticlePubMedPDF
- Cuffini AM, De Renzi G, Tullio V, Paizis G, Carlone NA. 1996. Potentiation of human polymorphonuclear leukocyte phagocytosis and intracellular bactericidal activity by amoxycillin/clavulanic acid. Drugs Exp Clin Res. 22: 9–15. PubMed
- Dartois V. 2014. The path of anti-tuberculosis drugs: from blood to lesions to mycobacterial cells. Nat Rev Microbiol. 12: 159–167. ArticlePubMedPMCPDF
- Dartois V, Barry CE. 2013. A medicinal chemists’ guide to the unique difficulties of lead optimization for tuberculosis. Bioorg Med Chem Lett. 23: 4741–4750. ArticlePubMedPMC
- De Francesco V, Bellesia A, Ridola L, Manta R, Zullo A. 2017. First-line therapies for Helicobacter pylori eradication: a critical reappraisal of updated guidelines. Ann Gastroenterol. 30: 373–379. ArticlePubMedPMC
- Domalaon R, Idowu T, Zhanel GG, Schweizer F. 2018. Antibiotic hybrids: the next generation of agents and adjuvants against Gram-negative pathogens? Clin Microbiol Rev. 31: e00077-17.ArticlePubMedPMCLink
- Etayash H, Alford M, Akhoundsadegh N, Drayton M, Straus SK, et al. 2021. Multifunctional antibiotic-host defense peptide conjugate kills bacteria, eradicates biofilms, and modulates the innate immune response. J Med Chem. 64: 16854–16863. ArticlePubMedLink
- Finlay J, Miller L, Poupard JA. 2003. A review of the antimicrobial activity of clavulanate. J Antimicrob Chemother. 52: 18–23. ArticlePubMed
- Fratoni AJ, Mah JW, Nicolau DP, Kuti JL. 2022. Imipenem/cilastatin/relebactam pharmacokinetics in critically ill patients with augmented renal clearance. J Antimicrob Chemother. 77: 2992–2999. ArticlePubMedPDF
- Gambato S, Bellotto O, Mardirossian M, Di Stasi A, Gennaro R, et al. 2023. Designing new hybrid antibiotics: proline-rich antimicrobial peptides conjugated to the aminoglycoside tobramycin. Bioconjugate Chem. 34: 1212–1220. ArticleLink
- Gao ML, Kotsogianni I, Skoulikopoulou F, Brüchle NC, Innocenti P, et al. 2024. Synthesis and evaluation of carbapenem/metallo-β-lactamase inhibitor conjugates. ChemMedChem. 19: e202400302. ArticlePubMed
- Gerding DN, Cornely OA, Grill S, Kracker H, Marrast AC, et al. 2019. Cadazolid for the treatment of Clostridium difficile infection: results of two double-blind, placebo-controlled, non-inferiority, randomised phase 3 trials. Lancet Infect Dis. 19: 265–274. ArticlePubMed
- Gill EE, Franco OL, Hancock REW. 2015. Antibiotic adjuvants: diverse strategies for controlling drug-resistant pathogens. Chem Biol Drug Des. 85: 56–78. ArticlePubMedLink
- Gómez-Lus ML, Aguilar L, Martín M, Giménez MJ, Martínez P, et al. 1997. Intracellular and extracellular killing of a penicillin-resistant, serotype-9 strain of Streptococcus pneumoniae by polymorphonuclear leucocytes in the presence of sub-inhibitory concentrations of clavulanic acid. J Antimicrob Chemother. 40: 142–144. ArticlePubMed
- Goncette V, Layios N, Descy J, Frippiat F. 2021. Continuous infusion, therapeutic drug monitoring and outpatient parenteral antimicrobial therapy with ceftazidime/avibactam: a retrospective cohort study. J Glob Antimicrob Resist. 26: 15–19. ArticlePubMed
- Guo Y, Huang C, Su H, Zhang Z, Chen M, et al. 2022. Luteolin increases susceptibility to macrolides by inhibiting MsrA efflux pump in Trueperella pyogenes. Vet Res. 53: 3.ArticlePubMedPMCPDF
- Hamilton-Miller JMT, Newton GGF, Abraham EP. 1970. Products of aminolysis and enzymic hydrolysis of the cephalosporins. Biochem J. 116: 371–384. ArticlePubMedPMCPDF
- Hardie KR. 2020. Antimicrobial resistance: the good, the bad, and the ugly. Emerg Top Life Sci. 4: 129–136. ArticlePubMedPDF
- Hegde SS, Okusanya OO, Skinner R, Shaw JP, Obedencio G, et al. 2012. Pharmacodynamics of TD-1792, a novel glycopeptide-cephalosporin heterodimer antibiotic used against Gram-positive bacteria, in a neutropenic murine thigh model. Antimicrob Agents Chemother. 56: 1578–1583. ArticlePubMedPMCLink
- Jones RN, Barry AL, Thornsberry C. 1989. Antimicrobial activity of Ro 23-9424, a novel ester-linked codrug of fleroxacin and desacetylcefotaxime. Antimicrob Agents Chemother. 33: 944–950. ArticlePubMedPMCLink
- Kadioglu A, Gingles NA, Grattan K, Kerr A, Mitchell TJ, et al. 2000. Host cellular immune response to pneumococcal lung infection in mice. Infect Immun. 68: 492–501. ArticlePubMedPMCLink
- Kaur K, Gupta A, Sharma A, Walia G, Singh B, et al. 2014. Evaluation of efficacy and tolerability of cefotaxime and sulbactam versus cefepime and tazobactam in patients of urinary tract infection—a prospective comparative study. J Clin Diagn Res. 8: HC05–HC08. Article
- Kaye KS, Belley A, Barth P, Lahlou O, Knechtle P, et al. 2022. Effect of cefepime/enmetazobactam vs piperacillin/tazobactam on clinical cure and microbiological eradication in patients with complicated urinary tract infection or acute pyelonephritis: a randomized clinical trial. JAMA. 328: 1304–1314. ArticlePubMedPMC
- Kaye KS, Bhowmick T, Metallidis S, Bleasdale SC, Sagan OS, et al. 2018. Effect of meropenem-vaborbactam vs piperacillin-tazobactam on clinical cure or improvement and microbial eradication in complicated urinary tract infection: the TANGO I randomized clinical trial. JAMA. 319: 788–799. ArticlePubMedPMC
- Kaye KS, Shorr AF, Wunderink RG, Du B, Poirier GE, et al. 2023. Efficacy and safety of sulbactam-durlobactam versus colistin for the treatment of patients with serious infections caused by Acinetobacter baumannii-calcoaceticus complex: a multicentre, randomised, active-controlled, phase 3, non-inferiority clinical trial. Lancet Infect Dis. 23: 1072–1084. ArticlePubMed
- Kerantzas CA, Jacobs WR. 2017. Origins of combination therapy for tuberculosis: lessons for future antimicrobial development and application. mBio. 8: e01586-16.ArticlePubMedPMCLink
- Khan IA, Mirza ZM, Kumar A, Verma V, Qazi GN. 2006. Piperine, a phytochemical potentiator of ciprofloxacin against Staphylococcus aureus. Antimicrob Agents Chemother. 50: 810–812. ArticlePubMedPMCLink
- Kim DY, Kim HJ. 2021. Function of fimsbactin B as an Acinetobacter-selective antibiotic delivery vehicle. Org Lett. 23: 5256–5260. Article
- Lai Y-H, Franke R, Pinkert L, Overwin H, Brönstrup M. 2023. Molecular signatures of the eagle effect induced by the artificial siderophore conjugate LP-600 in E. coli. ACS Infect Dis. 9: 567–581. ArticlePubMedPMCLink
- Larkins-Ford J, Aldridge BB. 2022. Advances in the design of combination therapies for the treatment of tuberculosis. Expert Opin Drug Discov. 18: 83–97. ArticlePubMedPMC
- Laws M, Shaaban A, Rahman KM. 2019. Antibiotic resistance breakers: current approaches and future directions. FEMS Microbiol Rev. 43: 490–516. ArticlePubMedPMCPDF
- Lenaerts A, Barry CE, Dartois V. 2015. Heterogeneity in tuberculosis pathology, microenvironments and therapeutic responses. Immunol Rev. 264: 288–307. ArticlePubMedPMCLink
- Lepper MH, Dowling HF. 1951. Treatment of pneumococcic meningitis with penicillin compared with plus aureomycin: studies including observations on an apparent antagonism between penicillin and aureomycin. AMA Arch Intern Med. 8: 489–494.
- Leuthner KD, Vidaillac C, Cheung CM, Rybak MJ. 2010. In vitro activity of the new multivalent glycopeptide-cephalosporin antibiotic TD-1792 against vancomycin-nonsusceptible Staphylococcus isolates. Antimicrob Agents Chemother. 54: 3799–3803. ArticlePubMedPMCLink
- Li X, Liu Y, Wang M, Gao L, Liu J, et al. 2024. Safety, pharmacokinetics, and efficacy of rifasutenizol, a novel dual-targeted antibacterial agent in healthy participants and patients in China with Helicobacter pylori infection: four randomised clinical trials. Lancet Infect Dis. 24: 650–664. ArticlePubMed
- Liu F, Kou Q, Li H, Cao Y, Chen M, et al. 2024. Discovery of YFJ-36: design, synthesis, and antibacterial activities of catechol-conjugated β-lactams against Gram-negative bacteria. J Med Chem. 67: 6705–6725. ArticlePubMed
- Liu R, Miller PA, Miller MJ. 2021. Conjugation of aztreonam, a synthetic monocyclic β-lactam antibiotic, to a siderophore mimetic significantly expands activity against Gram-negative bacteria. ACS Infect Dis. 7: 2979–2986. ArticlePubMedPMC
- Liu R, Miller PA, Vakulenko SB, Stewart NK, Boggess WC, et al. 2018. A synthetic dual drug sideromycin induces Gram-negative bacteria to commit suicide with a Gram-positive antibiotic. J Med Chem. 61: 3845–3854. ArticlePubMed
- Ljungquist O, Magda M, Giske CG, Tellapragada C, Nazarchuk O, et al. 2024. Pandrug-resistant Klebsiella pneumoniae isolated from Ukrainian war victims are hypervirulent. J Infect. 89: 106312.ArticlePubMed
- Locher HH, Caspers P, Bruyère T, Schroeder S, Pfaff P, et al. 2014a. Investigations of the mode of action and resistance development of cadazolid, a new antibiotic for treatment of Clostridium difficile infections. Antimicrob Agents Chemother. 58: 901–908. ArticleLink
- Locher HH, Seiler P, Chen X, Schroeder S, Pfaff P, et al. 2014b. In vitro and in vivo antibacterial evaluation of cadazolid, a new antibiotic for treatment of Clostridium difficile infections. Antimicrob Agents Chemother. 58: 892–900. ArticleLink
- Lomovskaya O, Warren MS, Lee A, Galazzo J, Fronko R, et al. 2001. Identification and characterization of inhibitors of multidrug resistance efflux pumps in Pseudomonas aeruginosa: novel agents for combination therapy. Antimicrob Agents Chemother. 45: 105–116. ArticlePubMedPMCLink
- Long DD, Aggen JB, Christensen BG, Judice JK, Hegde SS, et al. 2008. A multivalent approach to drug discovery for novel antibiotics. J Antibiot. 61: 595–602. ArticlePDF
- Louie T, Nord CE, Talbot GH, Wilcox M, Gerding DN, et al. 2015. Multicenter, double-blind, randomized, phase 2 study evaluating the novel antibiotic cadazolid in patients with Clostridium difficile infection. Antimicrob Agents Chemother. 59: 6266–6273. ArticlePubMedPMCLink
- Loupias P, Laumaillé P, Morandat S, Mondange L, Guillier S, et al. 2023. Synthesis and study of new siderophore analog-ciprofloxacin conjugates with antibiotic activities against Pseudomonas aeruginosa and Burkholderia spp. Eur J Med Chem. 245: 114921.ArticlePubMed
- Lucasti C, Vasile L, Sandesc D, Venskutonis D, McLeroth P, et al. 2016. Phase 2, dose-ranging study of relebactam with imipenem-cilastatin in subjects with complicated intra-abdominal infection. Antimicrob Agents Chemother. 60: 6234–6243. ArticlePubMedPMCLink
- Ma Z, He S, Yuan Y, Zhuang Z, Liu Y, et al. 2022. Design, synthesis, and characterization of TNP-2198, a dual-targeted rifamycin-nitroimidazole conjugate with potent activity against microaerophilic and anaerobic bacterial pathogens. J Med Chem. 65: 4481–4495. ArticlePubMedPMC
- Ma Z, Lynch AS. 2016. Development of a dual-acting antibacterial agent (TNP-2092) for the treatment of persistent bacterial infections. J Med Chem. 59: 6645–6657. ArticlePubMed
- Marinacci B, D’Agostino I, Angeli A, Carradori S, Melfi F, et al. 2024. Inhibition of Pseudomonas aeruginosa carbonic anhydrases, exploring ciprofloxacin functionalization toward new antibacterial agents: an in-depth multidisciplinary study. J Med Chem. 67: 19077–19102. ArticlePubMed
- Martín M, Gómez-Lus ML, Aguilar L, Martínez P, Giménez MJ, et al. 1997. Effect of clavulanic acid and/or polymorphonuclear neutrophils on amoxicillin bactericidal activity against Streptococcus pneumoniae. Eur J Clin Microbiol Infect Dis. 16: 512–516. ArticlePubMedPDF
- McKenna M. 2020. The antibiotic paradox: why companies can’t afford to create life-saving drugs. Nature. 584: 338–341. ArticlePubMedPDF
- Melander RJ, Melander C. 2017. The challenge of overcoming antibiotic resistance: an adjuvant approach? ACS Infect Dis. 3: 559–563. ArticlePubMedPMC
- Motz RN, Guo C, Sargun A, Walker GT, Sassone-Corsi M, et al. 2024. Conjugation to native and nonnative triscatecholate siderophores enhances delivery and antibacterial activity of a β-lactam to Gram-negative bacterial pathogens. J Am Chem Soc. 146: 7708–7722. ArticlePubMedPMCLink
- Moya B, Barcelo IM, Bhagwat S, Patel M, Bou G, et al. 2017. WCK 5107 (zidebactam) and WCK 5153 are novel inhibitors of PBP2 showing potent “β-lactam enhancer” activity against Pseudomonas aeruginosa, including multidrug-resistant metallo-β-lactamase-producing high-risk clones. Antimicrob Agents Chemother. 61: e02529-16.ArticlePubMedPMCLink
- Murray CJ, Ikuta KS, Sharara F, Swetschinski L, Robles Aguilar G, et al. 2022. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 399: 629–655. ArticlePubMedPMC
- Naghavi M, Vollset SE, Ikuta KS, Swetschinski LR, Gray AP, et al. 2024. Global burden of bacterial antimicrobial resistance 1990–2021: a systematic analysis with forecasts to 2050. Lancet. 404: 1199–1226. ArticlePubMedPMC
- Nichols WW, Stone GG, Newell P, Broadhurst H, Wardman A, et al. 2018. Ceftazidime-avibactam susceptibility breakpoints against Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob Agents Chemother. 62: e02590-17.ArticlePubMedPMCLink
- O’Callaghan CH, Kirby SM, Morris A, Waller RE, Duncombe RE. 1972. Correlation between hydrolysis of the β-lactam bond of the cephalosporin nucleus and expulsion of the 3-substituent. J Bacteriol. 110: 988–991. ArticlePubMedPMCLink
- O’Callaghan CH, Sykes RB, Staniforth SE. 1976. A new cephalosporin with a dual mode of action. Antimicrob Agents Chemother. 10: 245–248. ArticlePubMedPMCLink
- O’Neill AJA, Cove JH, Chopra I. 2001. Mutation frequencies for resistance to fusidic acid and rifampicin in Staphylococcus aureus. J Antimicrob Chemother. 47: 647–650. ArticlePubMed
- Osei Sekyere J, Amoako DG. 2017. Carbonyl cyanide m-chlorophenylhydrazone (CCCP) reverses resistance to colistin, but not to carbapenems and tigecycline, in multidrug-resistant Enterobacteriaceae. Front Microbiol. 8: 228.ArticlePubMedPMC
- Pascual A, Martínez LM, Aragón J, Perea EJ. 1989. Effect of amoxycillin and clavulanic acid, alone and in combination, on human polymorphonuclear leukocyte function against Staphylococcus aureus. Eur J Clin Microbiol Infect Dis. 8: 277–281. ArticlePubMedPDF
- Petrosillo N, Ioannidou E, Falagas M. 2008. Colistin monotherapy vs. combination therapy: evidence from microbiological, animal and clinical studies. Eur J Clin Microbiol Infect Dis. 14: 816–827. Article
- Pinkert L, Lai YH, Peukert C, Hotop SK, Karge B, et al. 2021. Antibiotic conjugates with an artificial MECAM-based siderophore are potent agents against Gram-positive and Gram-negative bacterial pathogens. J Med Chem. 64: 15440–15460. ArticlePubMedLink
- Plackett B. 2020. Why big pharma has abandoned antibiotics. Nature. 586: S50–S52. ArticlePDF
- Prasad NK, Seiple IB, Cirz RT, Rosenberg OS. 2022. Leaks in the pipeline: a failure analysis of Gram-negative antibiotic development from 2010 to 2020. Antimicrob Agents Chemother. 66: e00054-22.ArticlePubMedPMCLink
- Rahn HP, Liu X, Chosy MB, Sun J, Cegelski L, et al. 2024. Biguanide-vancomycin conjugates are effective broad-spectrum antibiotics against actively growing and biofilm-associated Gram-positive and Gram-negative ESKAPE pathogens and mycobacteria. J Am Chem Soc. 146: 22541–22552. ArticlePubMedPMCLink
- Rodríguez D, Lence E, Vázquez-Ucha JC, Beceiro A, González-Bello C. 2024. Novel penicillin-based sulfone-siderophore conjugates for restoring β-lactam antibiotic efficacy. ACS Omega. 9: 26484–26494. ArticlePubMedPMCLink
- Rueedi G, Panchaud P, Friedli A, Specklin JL, Hubschwerlen C, et al. 2024. Discovery and structure -activity relationship of cadazolid: a first-in-class quinoxolidinone antibiotic for the treatment of Clostridioides difficile infection. J Med Chem. 67: 9465–9484. ArticlePubMed
- Samir M, Ramadan M, Abdelrahman MH, Elbastawesy MA, Halby HM, et al. 2022. New potent ciprofloxacin-uracil conjugates as DNA gyrase and topoisomerase IV inhibitors against methicillin-resistant Staphylococcus aureus. Bioorg Med Chem. 73: 117004.ArticlePubMed
- Sargun A, Johnstone TC, Zhi H, Raffatellu M, Nolan EM. 2021. Enterobactin- and salmochelin-β-lactam conjugates induce cell morphologies consistent with inhibition of penicillin-binding proteins in uropathogenic Escherichia coli CFT073. Chem Sci. 12: 4041–4056. ArticlePubMedPMC
- Sato T, Yamawaki K. 2019. Cefiderocol: discovery, chemistry, and in vivo profiles of a novel siderophore cephalosporin. Clin Infect Dis. 69: S538–S543. ArticlePubMedPMCPDF
- Schumacher A, Steinke P, Bohnert JA, Akova M, Jonas D, et al. 2006. Effect of 1-(1-naphthylmethyl)-piperazine, a novel putative efflux pump inhibitor, on antimicrobial drug susceptibility in clinical isolates of Enterobacteriaceae other than Escherichia coli. J Antimicrob Chemother. 57: 344–348. ArticlePubMed
- Shi W, Chen F, Zou X, Jiao S, Wang S, et al. 2021. Design, synthesis, and antibacterial evaluation of vancomycin-LPS binding peptide conjugates. Bioorg Med Chem Lett. 45: 128122.ArticlePubMed
- Siedentop B, Kachalov VN, Witzany C, Egger M, Kouyos RD, et al. 2024. The effect of combining antibiotics on resistance: a systematic review and meta-analysis. eLife. 13: RP93740.ArticlePubMedPMCPDF
- Silver LL. 2011. Challenges of antibacterial discovery. Clin Microbiol Rev. 24: 71–109. ArticlePubMedPMCLink
- Singh R, Dwivedi S, Gaharwar U, Meena R, Rajamani P, et al. 2019. Recent updates on drug resistance in Mycobacterium tuberculosis. J Appl Microbiol. 128: 1547–1567. ArticlePubMedLink
- Singh S, Kalia NP, Joshi P, Kumar A, Sharma PR, et al. 2017. Boeravinone B, a novel dual inhibitor of the NorA bacterial efflux pump of Staphylococcus aureus and human P-glycoprotein, reduces biofilm formation and intracellular invasion of bacteria. Front Microbiol. 8: 1868.ArticlePubMedPMC
- Song Z, Zhou L, Wang W, Lan C, Tang T, et al. 2025. Rifasutenizol-based triple therapy versus bismuth plus clarithromycin-based triple therapy for first-line treatment of Helicobacter pylori infection in China (EVEREST-HP): a phase 3, multicentre, randomised, triple-dummy, double-blind, controlled, non-inferiority trial. Lancet Infect Dis. 26: 101–110. ArticlePubMed
- Soriano A, Montravers P, Bassetti M, Klyasova G, Daikos G, et al. 2023. The use and effectiveness of ceftazidime-avibactam in real-world clinical practice: EZTEAM study. Infect Dis Ther. 12: 891–917. ArticlePubMedPMCPDF
- Stryjewski ME, Potgieter PD, Li YP, Barriere SL, Churukian A, et al. 2012. TD-1792 versus vancomycin for treatment of complicated skin and skin structure infections. Antimicrob Agents Chemother. 56: 5476–5483. ArticlePubMedPMCLink
- Tamma PD, Cosgrove SE, Maragakis LL. 2012. Combination therapy for treatment of infections with Gram-negative bacteria. Clin Microbiol Rev. 25: 450–470. ArticlePubMedPMCLink
- Tamma PD, Heil EL, Justo JA, Mathers AJ, Satlin MJ, et al. 2024. Infectious Diseases Society of America 2024 guidance on the treatment of antimicrobial-resistant Gram-negative infections. Clin Infect Dis. ciae403.Article
- Theuretzbacher U, Bush K, Harbarth S, Paul M, Rex JH, et al. 2020. Critical analysis of antibacterial agents in clinical development. Nat Rev Microbiol. 18: 286–298. ArticlePubMedPDF
- Thomas D, Wessel C. 2022. The state of innovation in antibacterial therapeutics. Biotechnology Innovation Organization, USA. Available from https://www.bio.org/sites/default/files/2022-02/The-State-of-Innovation-in-Antibacterial-Therapeutics.pdf.
- Tiberi S, Du Plessis N, Walzl G, Vjecha MJ, Rao M, et al. 2018. Tuberculosis: progress and advances in development of new drugs, treatment regimens, and host-directed therapies. Lancet Infect Dis. 18: e183–e198. Article
- van Haren MJ, Tehrani KHME, Kotsogianni I, Wade N, Brüchle NC, et al. 2021. Cephalosporin prodrug inhibitors overcome metallo-β-lactamase driven antibiotic resistance. Chem Eur J. 27: 3806–3811. ArticlePubMedLink
- Vargiu AV, Ruggerone P, Opperman TJ, Nguyen ST, Nikaido H. 2014. Molecular mechanism of MBX2319 inhibition of Escherichia coli AcrB multidrug efflux pump and comparison with other inhibitors. Antimicrob Agents Chemother. 58: 6224–6234. ArticlePubMedPMCLink
- Ventola CL. 2015. The antibiotic resistance crisis: part 1: causes and threats. P T. 40: 277–283. PubMedPMC
- Wallis RS, Maeurer M, Mwaba P, Chakaya J, Rustomjee R, et al. 2016. Tuberculosis—advances in development of new drugs, treatment regimens, host-directed therapies, and biomarkers. Lancet Infect Dis. 16: e34–e46. ArticlePubMed
- Wang J, Jiao H, Meng J, Qiao M, Du H, et al. 2019. Baicalin inhibits biofilm formation and the quorum-sensing system by regulating the MsrA drug efflux pump in Staphylococcus saprophyticus. Front Microbiol. 10: 2800.ArticlePubMedPMC
- Wang D, Xie K, Zou D, Meng M, Xie M. 2018. Inhibitory effects of silybin on the efflux pump of methicillin-resistant Staphylococcus aureus. Mol Med Rep. 18: 827–833. ArticlePubMedPMC
- Wang YY, Zhang XY, Zhong XL, Huang YJ, Lin J, et al. 2023. Design and synthesis of 3-hydroxy-pyridin-4(1H)-ones-ciprofloxacin conjugates as dual antibacterial and antibiofilm agents against Pseudomonas aeruginosa. J Med Chem. 66: 2169–2193. ArticlePubMed
- Woods RJ, Read AF. 2023. Combination antimicrobial therapy to manage resistance. Evol Med Public Health. 11: 185–186. ArticlePubMedPMCPDF
- World Health Organization. 2020. Global tuberculosis report 2020. World Health Organization, Geneva. Available from https://www.who.int/publications/i/item/9789240013131.
- Worthington RJ, Melander C. 2013. Combination approaches to combat multidrug-resistant bacteria. Trends Biotechnol. 31: 177–184. ArticlePubMedPMC
- Wunderink RG, Giamarellos-Bourboulis EJ, Rahav G, et al. 2018. Effect and safety of meropenem-vaborbactam versus best-available therapy in patients with carbapenem-resistant Enterobacteriaceae infections: the TANGO II randomized clinical trial. Infect Dis Ther. 7: 439–455. ArticlePubMedPMCPDF
- Xia J, Li Y, He C, Yong C, Wang L, et al. 2023. Synthesis and biological activities of oxazolidinone pleuromutilin derivatives as a potent anti-MRSA agent. ACS Infect Dis. 9: 1711–1729. ArticlePubMedLink
- Yaxley J, Chakravarty B. 2014. Helicobacter pylori eradication—an update on the latest therapies. Aust Fam Physician. 43: 301–305. PubMed
- Yousfi MM, El-zimaity HMT, Al-assi MT, Cole RA, Genta RM, et al. 1995. Metronidazole, omeprazole and clarithromycin: an effective combination therapy for Helicobacter pylori infection. Aliment Pharmacol Ther. 9: 209–212. ArticlePubMed
References
Supplementary Information
References
Citations

- Pioneering strategies for overcoming bacterial drug resistance
Byoung Sik Kim
Journal of Microbiology.2026; 64(3): e2603100. CrossRef
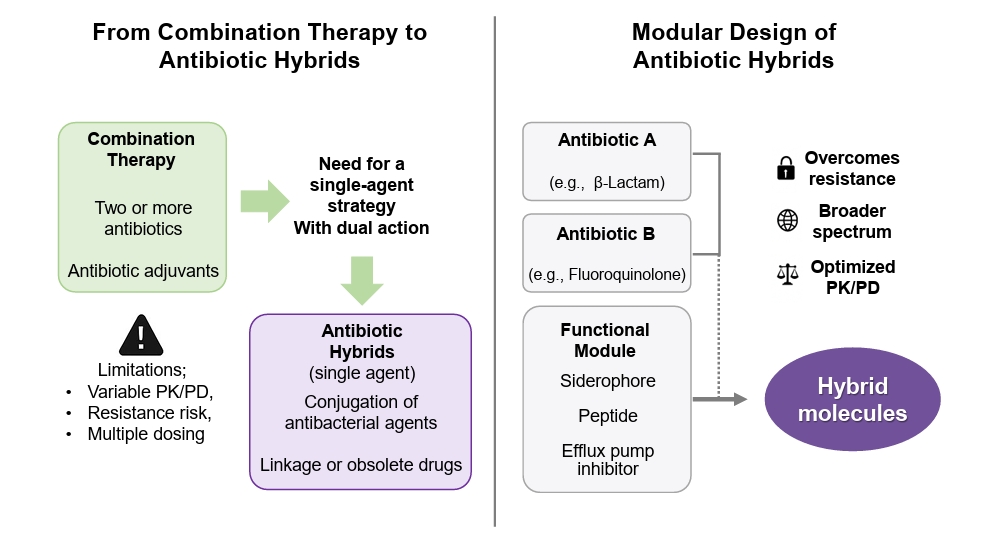
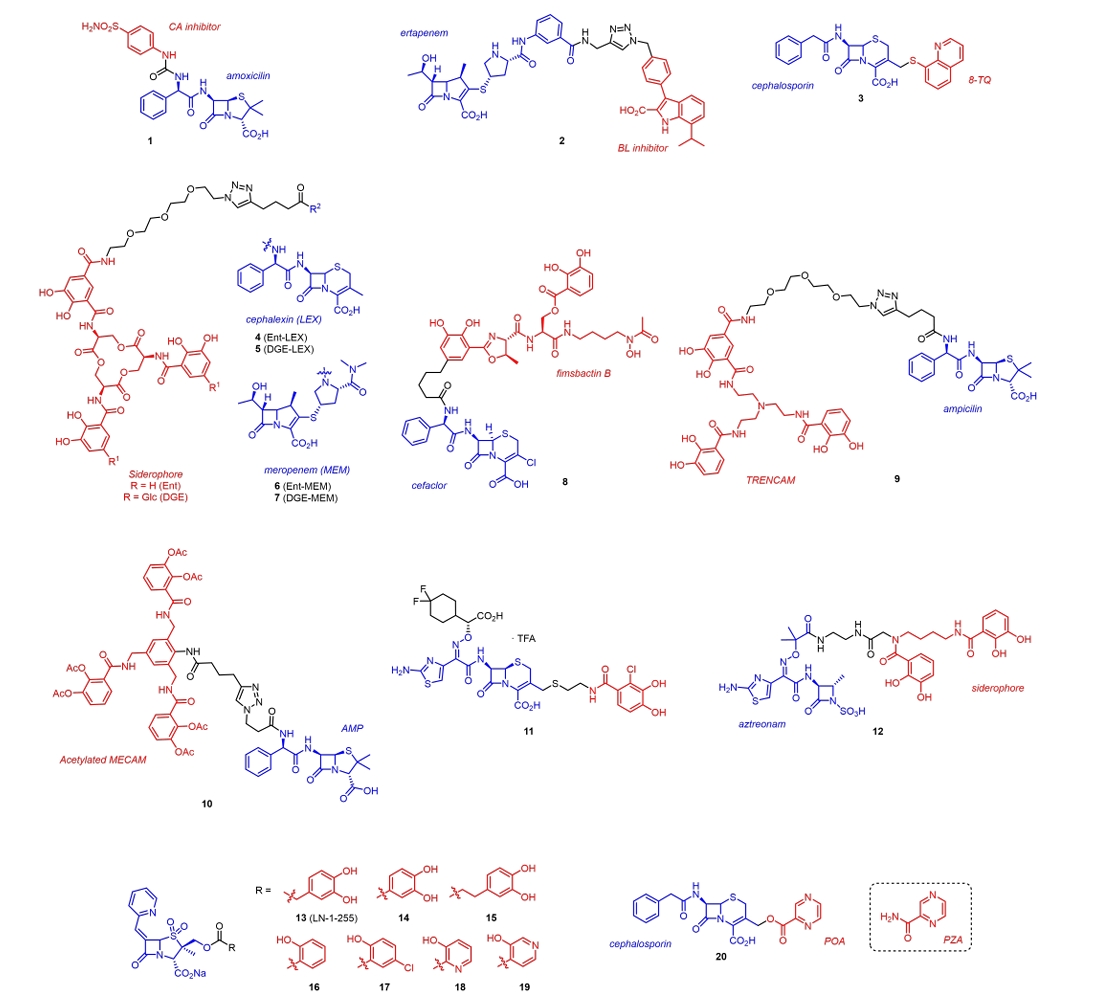
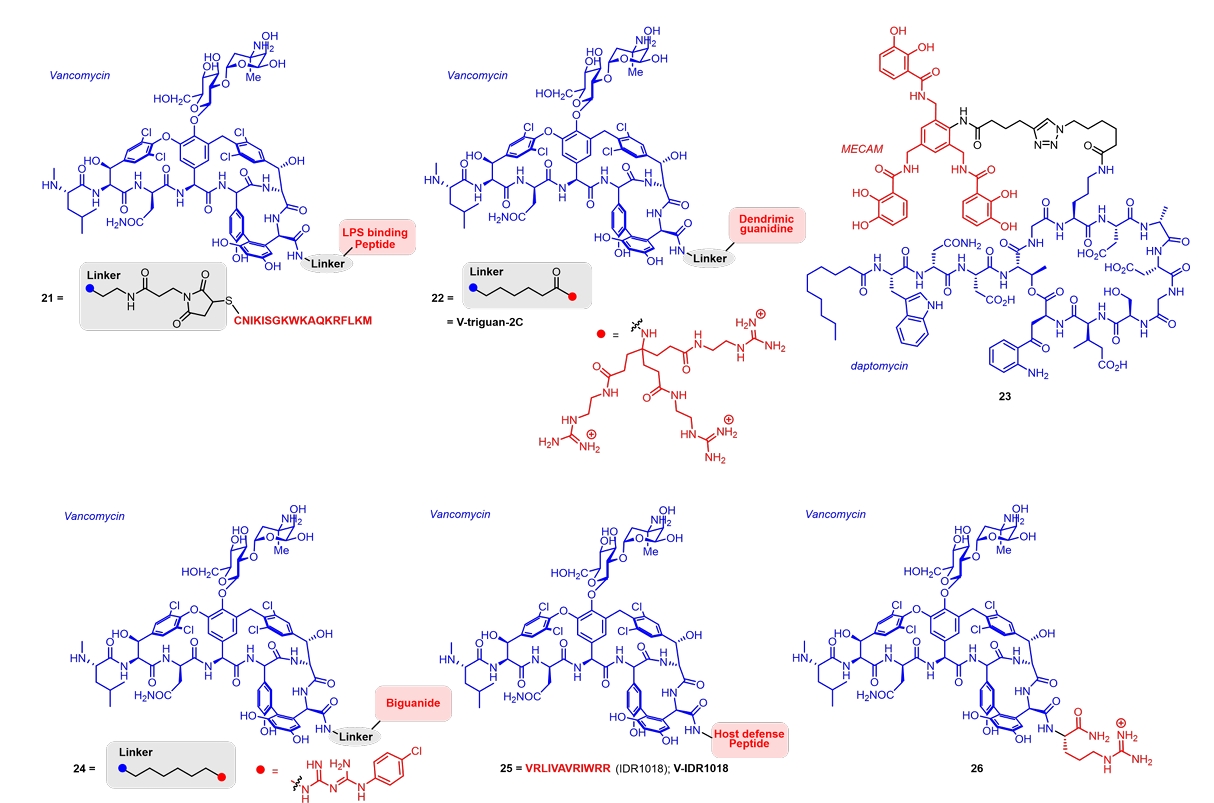
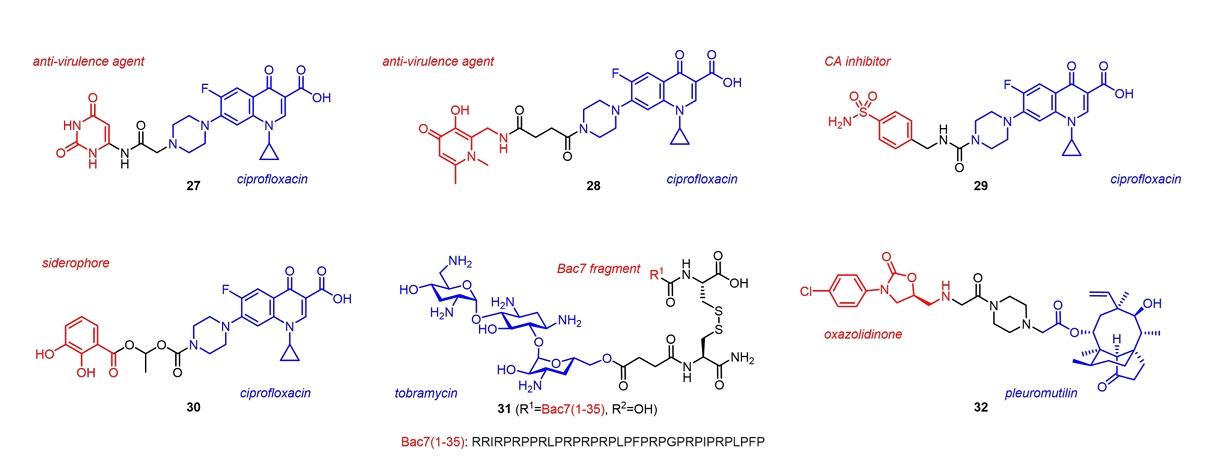
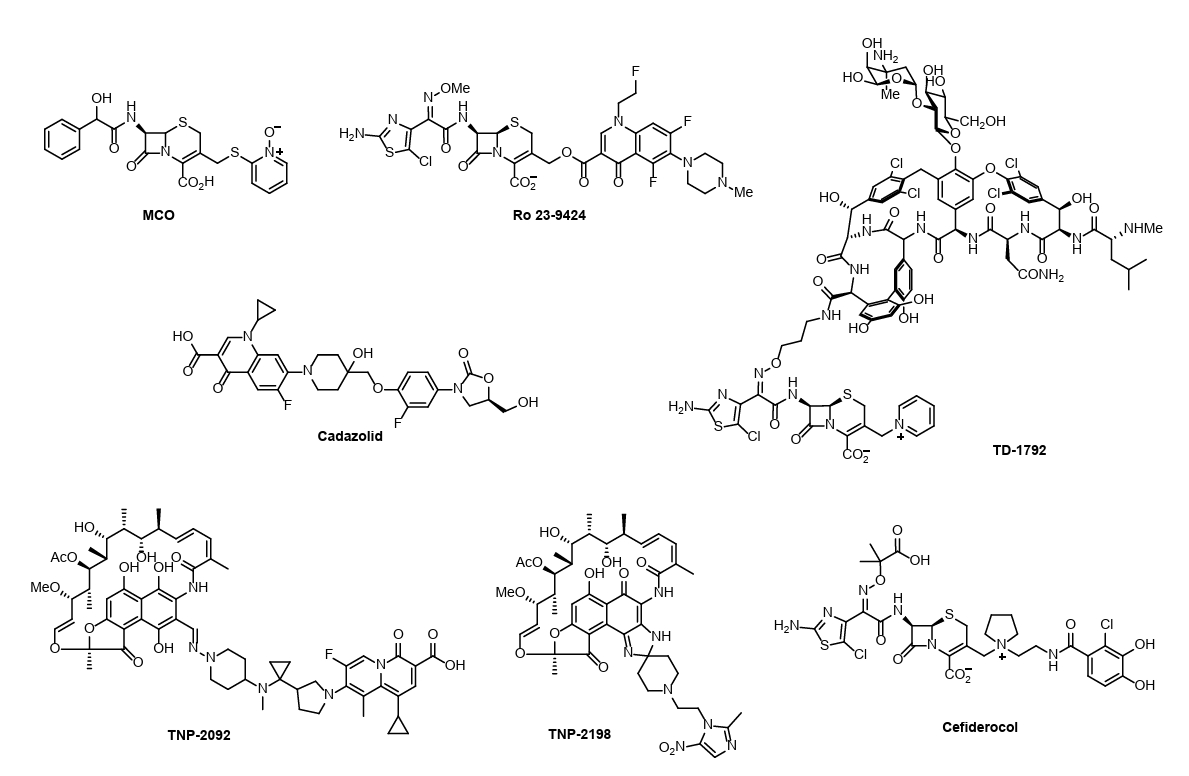
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
| Cpd. | Hybrid design | Target | Efficacy | Reference |
|---|---|---|---|---|
| 1 | Amoxicillin + CA inhibitor | N. gonorrhoeae (CDC-178) | MIC, 1 (1) vs 2 (amoxicillin) μg/ml | |
| 2 | Carbapenem + MBL Inhibitor | MBL Enzymes (NDM-1, IMP-1) | High Enzyme Inhibition (IC50 nM range), Inactive in cells | |
| 3 | Cephalosporin + 8-TQ | K. pneumoniae (MBL-producer) | Restored efficacy of Meropenem via enzyme-triggered release of 8-TQ. | |
| 6 | Meropenem + Enterobactin | E. coli (CFT073) | MIC, 0.01 (6) vs 0.1 (Meropenem) μM | |
| 8 | Cefaclor + Fimsbactin B | A. baumannii (ATCC 17978) | MIC, 0.5 (8) vs 64 (Cefaclor) μg/ml | |
| 9 | Ampicillin + TRENCAM | S. Typhimurium | MIC, 0.01 (9) vs 10 (Ampicillin) μM | |
| 10 | Ampicillin + MECAM | E. coli | MIC, 1.5 (10) vs 46 (Ampicillin) μM | |
| 11 | Cephalosporin + Catechol | A. baumannii (MDR Isolate) | MIC, 0.25 (11) vs ≥ 64 (Meropenem) μg/ml | |
| 20 | Cephalosporin + POA | M. tuberculosis (H37Rv ΔpncA) | MIC, 100 (20) vs > 800 (PZA) vs 100 (POA) μg/ml |
| Cpd. | Hybrid design | Target | Efficacy | Reference |
|---|---|---|---|---|
| 21 | Vancomycin + LPS-binding peptide | E. coli (AB1157) | MIC, 8 (21) vs > 88 (Vancomycin) μM | |
| 22 | Vancomycin + Dendrimeric guanidine | E. coli (UTI89) | MIC, 8 (22) vs 128 (Vancomycin) µM | |
| 23 | Daptomycin + MECAM | A. baumannii | MIC, 4.4 (23) vs > 39 (Daptomycin) µM | |
| 24 | Vancomycin + Biguanide | E. faecium (ATCC 51559) | MIC, 4 (24) vs 512 (Vancomycin) µM | |
| 25 | Vancomycin + IDR1018 peptide | MRSA biofilms | Minimal biofilm eradication conc. 16 (25) vs > 128 (Vancomycin) µg/ml | |
| 26 | Vancomycin + Arginine | M. abscessus (ATCC 19977) | MIC, 16 (26) vs 64 (Vancomycin) µM | |
| 27 | Ciprofloxacin + Uracil | MRSA (AUMC261) | MIC, 0.031 (27) vs 0.57 (Ciprofloxacin) µM | |
| 28 | Ciprofloxacin + Anti-virulence agent | P. aeruginosa Biofilms | Biofilm reduction at 1/4 MIC, 78.3% (28) vs 12.6% (Ciprofloxacin) | |
| 30 | Ciprofloxacin + Siderophore | B. pseudomallei (BPS2020IRBA003) | MIC, 2 (30) vs 16 (Ciprofloxacin) µg/ml | |
| 31 | Tobramycin + Bac7 peptide fragment | E. coli (BW25113) | MIC, 1 (31) vs 4 (Tobramycin) µM | |
| 32 | Pleuromutilin + Oxazolidinone | MRSA (ATCC33591) | MIC, 0.063 (32) vs > 0.5 (Pleuromutilin) μg/ml |
| Name (Year) | Hybrid design | Target | Current status | Features |
|---|---|---|---|---|
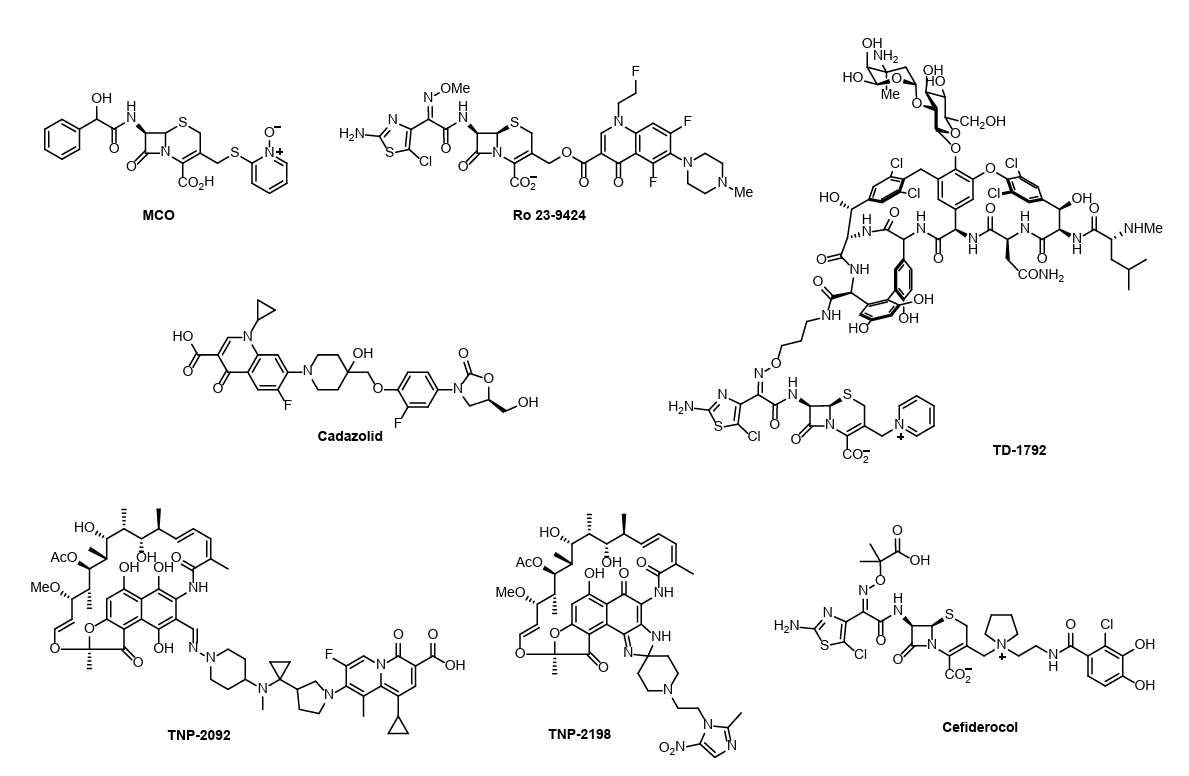
| MCO (1976) | Cephalosporin + Omadine | G(+) & G(-) | Discontinued | Demonstrated release of active omadine upon β-lactamase hydrolysis, but halted due to systemic toxicity |
| Ro 23-9424 (1990) | Desacetylcefotaxime (C-3) + Fleroxacin | G(+) & G(-) | Discontinued | Broad-spectrum ester-linked prodrug excreted intact. Discontinued due to lack of therapeutic advantage over co-administration. |
| TD-1792 (2008) | Vancomycin + Cephalosporin | G(+) (MRSA) | Phase 3 | Dual-targeting mechanism inhibiting both Lipid II and PBPs. Showed non-inferiority to vancomycin with high bactericidal activity in Phase II. |
| Cadazolid (2014) | Oxazolidinone + Fluoroquinolone | Clostridioides difficile infection (CDI) | Discontinued | Low systemic absorption ideal for CDI. Discontinued after failing to demonstrate non-inferiority in Phase III |
| TNP-2092 (2016) | Rifamycin + Quinolizinone | H. pylori, Biofilms | Phase 3 | Dual inhibition (RNA polymerase + Gyrase/Topo IV) minimizes resistance frequency. Effective against persistent biofilms. |
| Cefiderocol (2019) | Siderophore + Cephalosporin | MDR G(-) | Approved | Active transport via iron channels overcomes OM barrier. Stable against carbapenemases. |
| TNP-2198 (2022) | Rifamycin + Nitroimidazole | Anaerobes (H. pylori) | Phase 3 | Multi-targeting of microaerophiles. Recent trials confirmed high eradication rates in H. pylori triple therapy. |
Table 1.
Table 2.
Table 3.
TOP
 MSK
MSK
 ePub Link
ePub Link Cite this Article
Cite this Article






