- About
- Browse Articles
-
Special Issues
- Pioneering strategies for overcoming bacterial drug resistance (2026)
- Advancing microbial engineering through synthetic biology (2025)
- Host-associated microbiome (2024)
- Bacterial regulatory mechanisms for the control of complex cellular mechanisms (2023)
- Two years into COVID-19 pandemic: Where are we? (2022)
- Collections
- For Contributors
- Policies
- E-Submission

- About
- Browse Articles
-
Special Issues
- Pioneering strategies for overcoming bacterial drug resistance (2026)
- Advancing microbial engineering through synthetic biology (2025)
- Host-associated microbiome (2024)
- Bacterial regulatory mechanisms for the control of complex cellular mechanisms (2023)
- Two years into COVID-19 pandemic: Where are we? (2022)
- Collections
- Policies
- For Contributors
Articles
- Page Path
- HOME > J. Microbiol > Volume 64(2); 2026 > Article
-
Full article
Lacticaseibacillus paracasei KBL382 contributes to the immunomodulation in THP-1 cells - MinJoong Kim1,†, Min Jung Jo1,†, SungJun Park2,3,4, Seoung Bum Lee1, Sung Jae Jang1,4, Cheonghoon Lee1,5, Woon-Ki Kim1,5,6,*, GwangPyo Ko1,2,3,5,*
-
Journal of Microbiology 2026;64(2):e2509016.
DOI: https://doi.org/10.71150/jm.2509016
Published online: February 28, 2026
1Graduate School of Public Health, Seoul National University, Seoul 08826, Republic of Korea
2N-Bio, Seoul National University, Seoul 08826, Republic of Korea
3KoBioLabs, Inc., Seoul 08826, Republic of Korea
4weBiom Inc., Seoul 08826, Republic of Korea
5Institute of Health and Environment, Seoul National University, Seoul 08826, Republic of Korea
6Brain Korea for Global Leader of Better Environmental health (BK4GLOBE), Seoul National University, Seoul 08826, Republic of Korea
- *Correspondence Woon-ki Kim kimunki@hanmail.net GwangPyo Ko gko@snu.ac.kr
- †These authors contributed equally to this work.
© The Microbiological Society of Korea
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 706 Views
- 43 Download
ABSTRACT
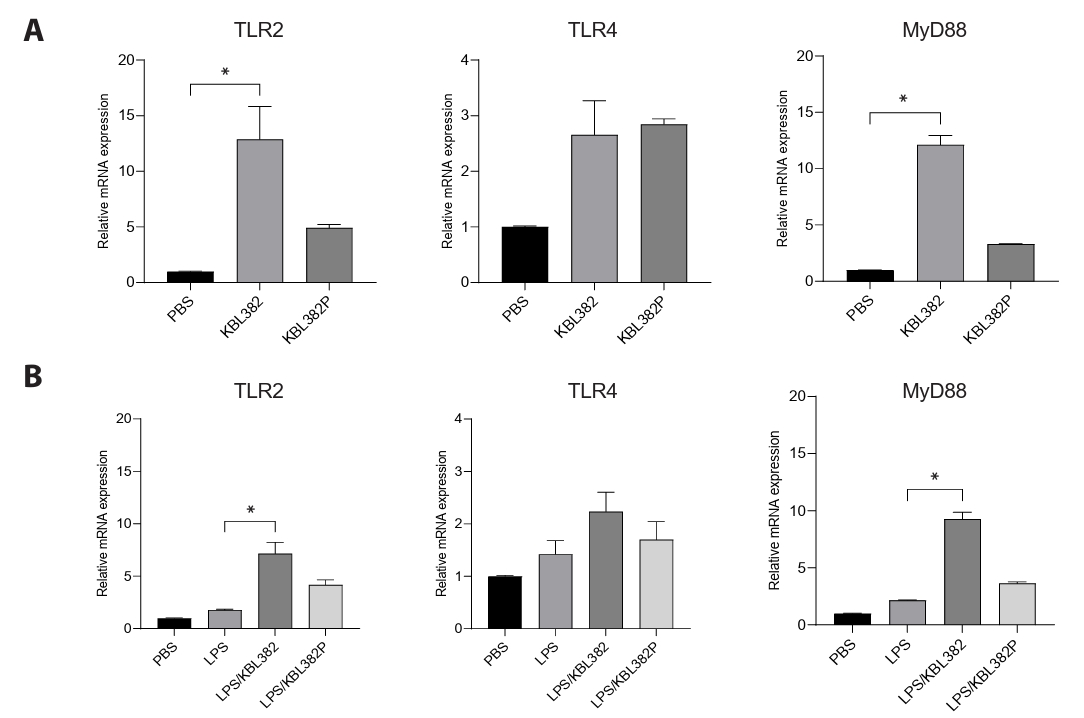
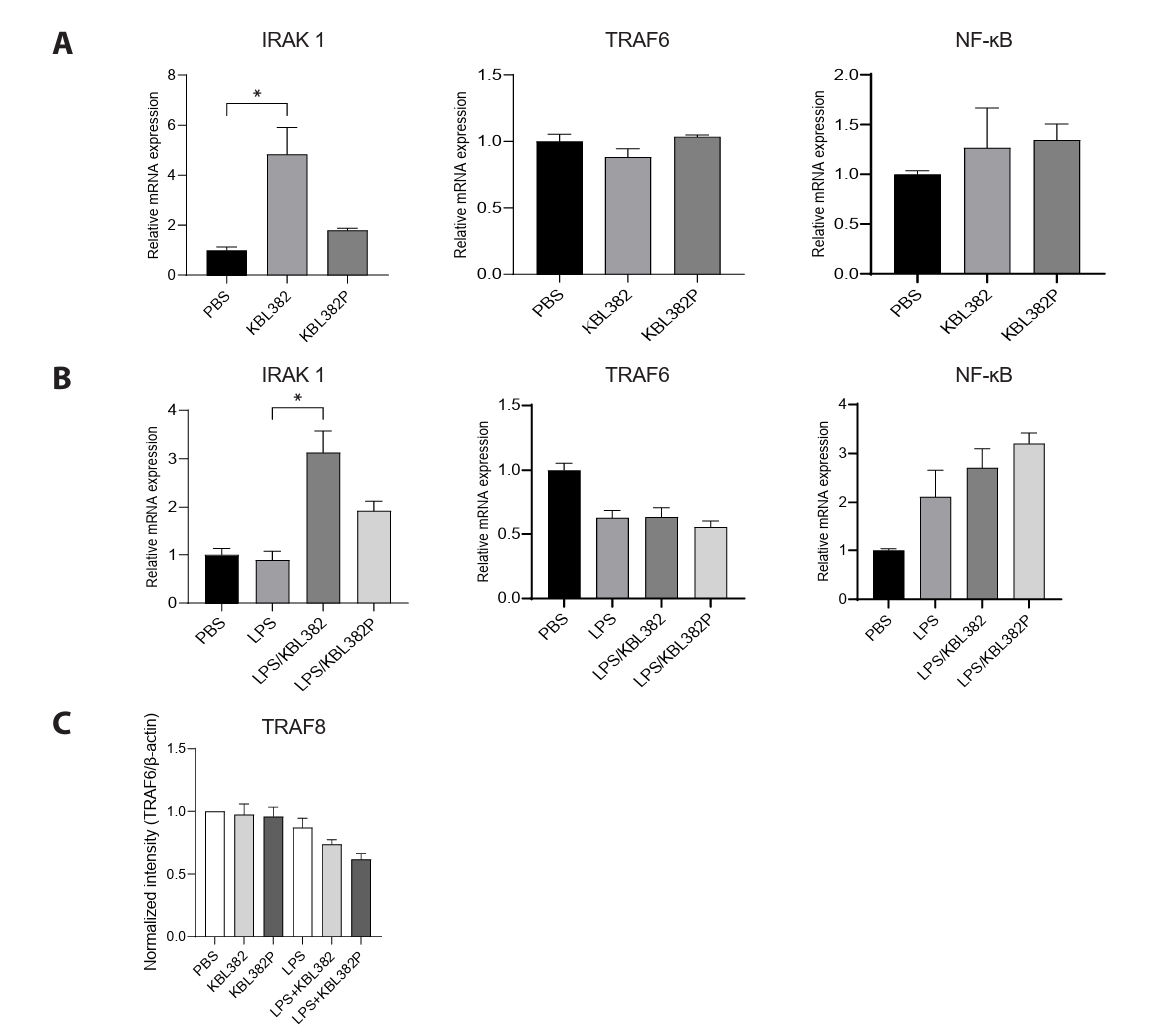
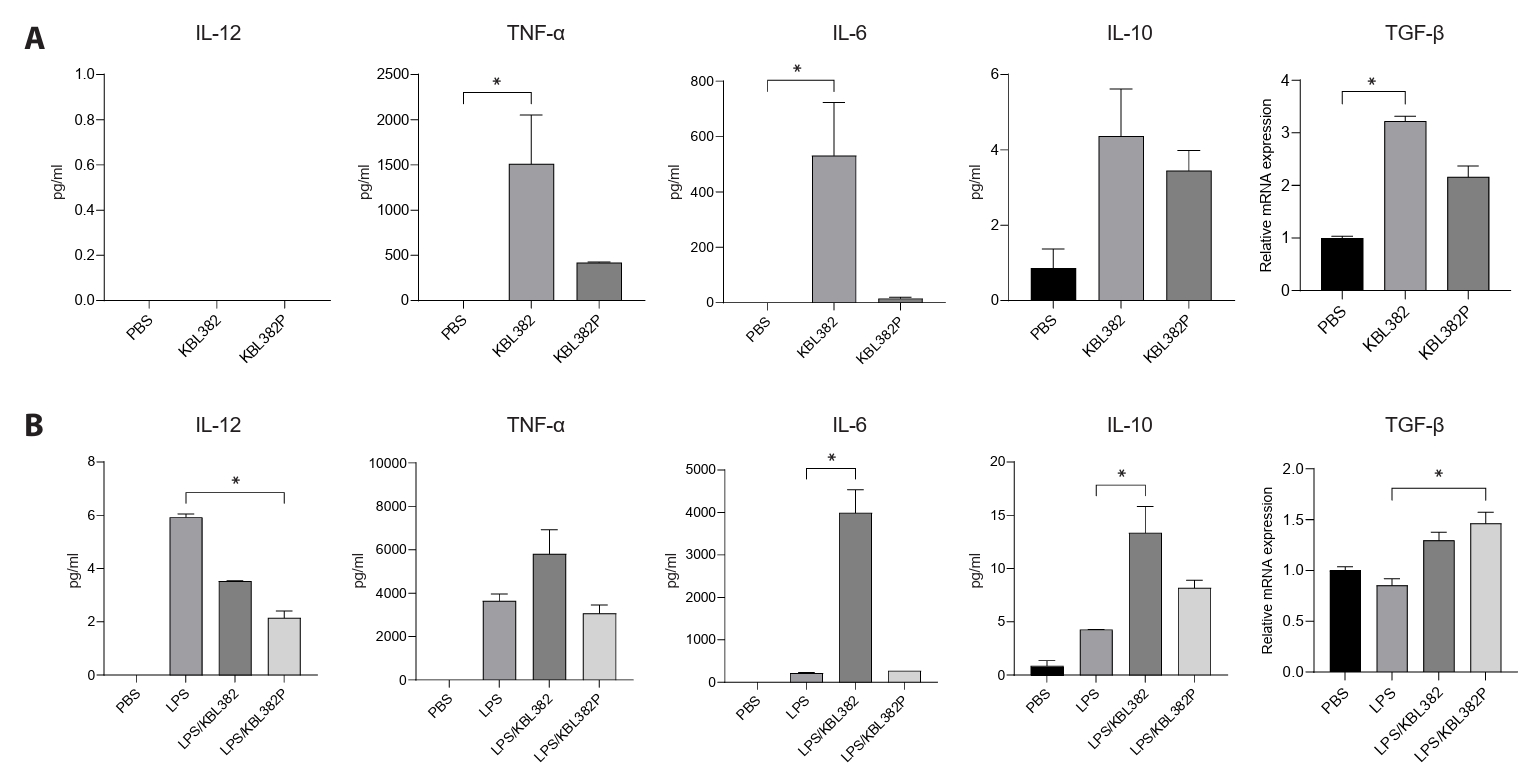
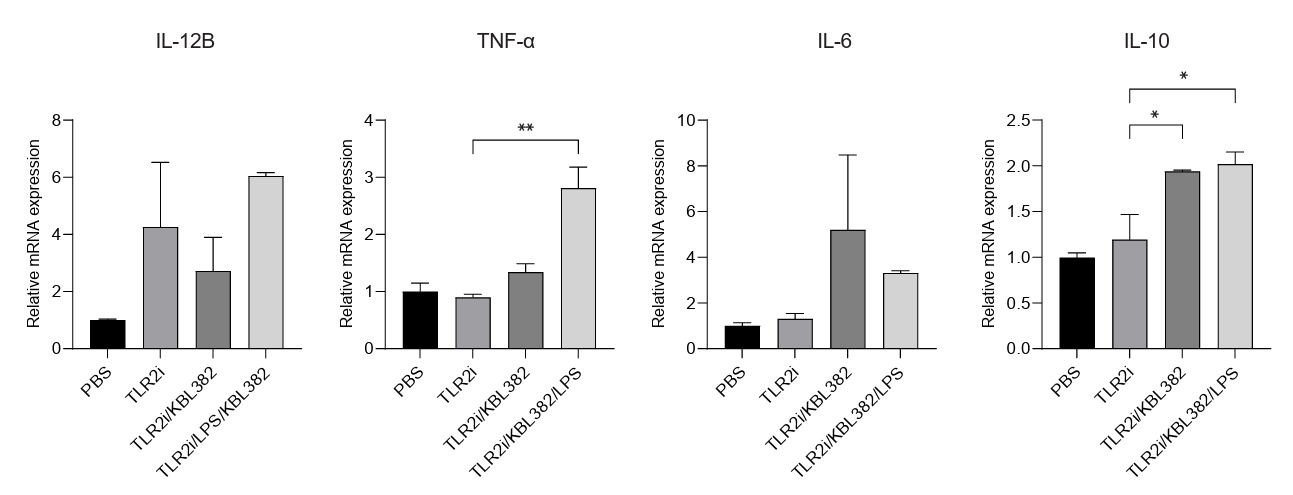
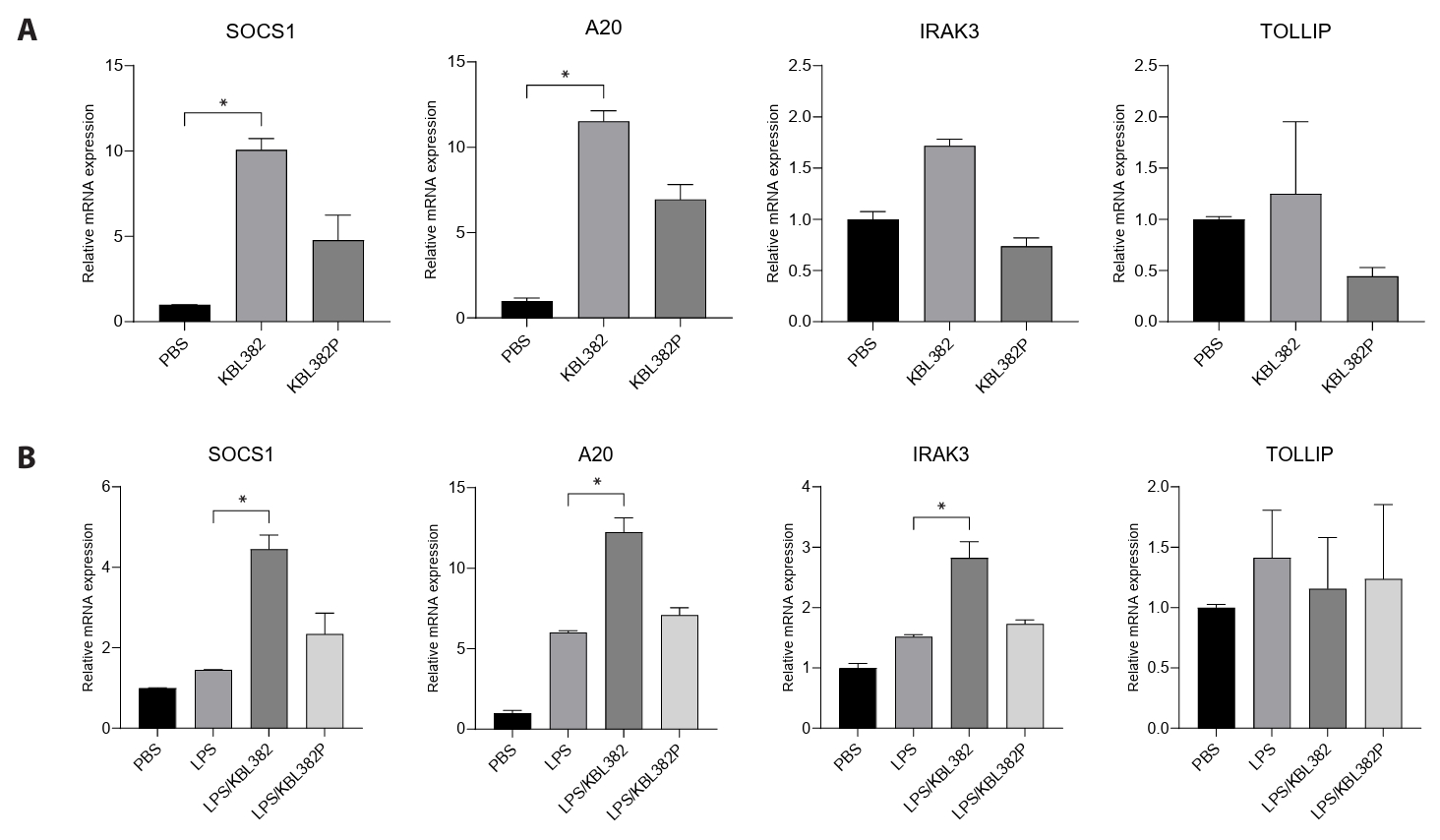
- Gut microbiome imbalance can induce inflammatory responses via Toll-like receptor 2 (TLR2) signaling pathways. Lactobacillus spp., popularly applied as probiotics in both humans and animals, have come into the spotlight for their strong immunomodulatory effects. We aimed to evaluate the immunomodulatory potential of live or pasteurized Lacticaseibacillus paracasei (L. paracasei) KBL382, isolated from healthy Korean individuals, in an in vitro monocytic THP-1 cell model. Live L. paracasei KBL382 significantly increased TLR2 and MyD88 expressions and induced IRAK1 expression, irrespective of lipopolysaccharide (LPS) stimulation (p < 0.05). Under LPS stimulation, THP-1 cells treated with live L. paracasei KBL382 showed significantly increased interleukin (IL)-6 and IL-10 levels (p < 0.05). Pasteurized L. paracasei exhibited a decrease in IL-12 levels (p < 0.05). Moreover, live L. paracasei KBL382 also markedly elevated A20 and SOCS1 expressions, the critical negative regulators of inflammation, regardless of LPS stimulation (p < 0.05). The expression of IRAK3, another negative regulator of inflammation, was increased in THP-1 cells with live L. paracasei KBL382 under LPS stimulation (p < 0.05). Our findings demonstrate that L. paracasei KBL382 contributes to the immunomodulation in THP-1 cells by coordinating both positive and negative regulatory signaling. L. paracasei KBL382 could be used as a promising probiotic strain for attenuating chronic inflammation through the gut-immune axis mechanisms.
Introduction
Materials and Methods
Results
Discussion
Acknowledgments
Open Access funding was enabled and organized by Seoul National University. This research was supported by the Basic Science Research Program through the National Research Foundation (NRF) funded by the Ministry of Education (no. NRF-2021R1I1A1A01048923) and the Bio & Medical Technology Development Program of the NRF funded by the Korean government (MSIT) (no. RS-2022-NR067344).
Conflict of Interest
G.K. is the chief executive officer of KoBioLabs, Inc. S.P. is an employee of KoBioLabs, Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability
All data supporting the findings of this study are available from the corresponding author upon reasonable request.
| Target | Sequence | Reference |
|---|---|---|
| A20 | Fw: 5’- AACATTTTGCTGCTGCCTC -3’ | Xiong et al. (2011) |
| Rv: 5’- AGGTGCTTTGTGTGGTTCG -3’ | ||
| GAPDH | Fw: 5’- GGAAGGTGAAGGTCGGAGTC -3’ | Han et al. (2020b) |
| Rv: 5’- TCAGCCTTGACGGTGCCATG -3’ | ||
| IL-6 | Fw: 5’- CATCCTCGACGGCATCTCAG -3’ | Kanmani and Kim (2018) |
| Rv: 5’- GCTCTGTTGCCTGGTCCTC -3’ | ||
| IL-10 | Fw: 5’- TCAGGGTGGCGACTCTAT -3’ | |
| Rv: 5’- TGGGCTTCTTCTAAATCGTTC -3’ | ||
| IL-12B | Fw: 5’- GGCTCCATGAAGGTGCTAC -3’ | Hor et al. (2014) |
| Rv: 5’- GTTCAGCCTCAGAATGCAAAA -3’ | ||
| IRAK1 | Fw: 5’- ACTGGCCCTTGGCAGCTC -3’ | Rahemi et al. (2019) |
| Rv: 5’- GGCCAGCTTCTGGACCATC -3’ | ||
| IRAK3 | Fw: 5’- TGCAACGCGGGCAAA -3’ | Sun et al. (2017) |
| Rv: 5’- TTTAGTGATGTGGGAGGATCTTCA -3’ | ||
| MyD88 | Fw: 5’- GAGCGTTTCGATGCTTCAT -3’ | Zarember and Godowski (2002) |
| Rv: 5’- CGGATCATCTCCTGCACAAA -3’ | ||
| NF-κB | Fw: 5’- TCAATGGCTACACAGGACCA -3’ | Ren et al. (2019) |
| Rv: 5’- CACTGTCACCTGGAAGCAGA -3’ | ||
| SOCS1 | Fw: 5’- CTGGGATGCCGTGTTATTTT-3’ | Lee et al. (2017) |
| Rv: 5’- TAGGAGGTGCGAGTTCAGGT-3’ | ||
| TLR2 | Fw: 5’- GCCAAAGTCTTGATTGATTGG-3’ | Erdinest et al. (2014) |
| Rv: 5’- TTGAAGTTCTCCAGCTCCTGG -3’ | ||
| TLR4 | Fw: 5’- GGTGGAAGTTGAACGAATGG -3’ | Asai et al. (2003) |
| Rv: 5’- CCAGCAAGAAGCATCAGGTG -3’ | ||
| TNF-α | Fw: 5’- TCTCGAACCCCGAGTGACAA -3’ | Ren et al. (2019) |
| Rv: 5’- TATCTCTCAGCTCCACGCCA -3’ | ||
| TOLLIP | Fw: 5’-AGGTGACAACTGTCTCCGTC-3’ | Sun et al. (2017) |
| Rv: 5’-GCCAACTTTGCCTGTACCAC-3’ | ||
| TRAF6 | Fw: 5’-CCTTTGGCAAATGTCATCTGTG-3’ | Shen et al. (2013) |
| Rv: 5’-CTCTGCATCTTTTCATGGCAAC-3’ | ||
| TGF-β | Fw: 5′-GAA GGC AGA GTT CAG GGT CTT-3′ | Kwon et al. (2010) |
| Rv: 5′-GGT TCC TGT CTT TGT GGT GAA-3′ |
- Asai Y, Jinno T, Ogawa T. 2003. Oral treponemes and their outer membrane extracts activate human gingival epithelial cells through toll-like receptor 2. Infect Immun. 71: 717–725. ArticlePubMedPMCLink
- Barroso FAL, de Jesus LCL, da Silva TF, Batista VL, Laguna J, et al. 2022. Lactobacillus delbrueckii CIDCA 133 ameliorates chemotherapy-induced mucositis by modulating epithelial barrier and TLR2/4/MyD88/NF-κB signaling pathway. Front Microbiol. 13: 858036.ArticlePubMedPMC
- Bermudez-Brito M, Plaza-Diaz J, Munoz-Quezada S, Gomez-Llorente C, Gil A. 2012. Probiotic mechanisms of action. Ann Nutr Metab. 61: 160–174. ArticlePubMedLink
- Chanput W, Mes JJ, Wichers HJ. 2014. THP-1 cell line: an in vitro cell model for immune modulation approach. Int Immunopharmacol. 23: 37–45. ArticlePubMed
- Duan T, Du Y, Xing C, Wang HY, Wang RF. 2022. Toll-like receptor signaling and its role in cell-mediated immunity. Front Immunol. 13: 812774.ArticlePubMedPMC
- Erdinest N, Aviel G, Moallem E, Anteby I, Yahalom C. 2014. Expression and activation of toll-like receptor 3 and toll-like receptor 4 on human corneal epithelial and conjunctival fibroblasts. J Inflamm. 11: 3.ArticlePubMedPMCPDF
- Fu J, Jia L, Wu L, Jiang Y, Zhao R, et al. 2024. Lactobacillus rhamnosus inhibits osteoclast differentiation by suppressing the TLR2/NF-κB pathway. Oral Dis. 4: 2373–2386. Article
- Han DH, Kim WK, Park S, Jang YJ, Ko G. 2020a. Lactobacillus paracasei treatment modulates mRNA expression in macrophages. Biochem Biophys Rep. 23: 100788.ArticlePubMedPMC
- Han MH, Lee JH, Kim G, Lee E, Lee YR. 2020b. Expression of the long noncoding RNA GAS5 correlates with liver fibrosis in patients with nonalcoholic fatty liver disease. Genes. 11: 545.ArticlePubMedPMC
- Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, et al. 2014. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 11: 506–514. ArticlePubMedPDF
- Hubbard LL, Moore BB. 2010. IRAK-M regulation and function in host defense and immune homeostasis. Infect Dis Rep. 2: e9.ArticlePubMedPMCLink
- Hor YT, Voon DC, Koo JK, Wang H, Lau WM, et al. 2014. A role for RUNX3 in inflammation-induced expression of IL23A in gastric epithelial cells. Cell Rep. 8: 50–58. ArticlePubMedPMC
- Kanmani P, Kim H. 2018. Protective effects of lactic acid bacteria against TLR4-induced inflammatory response in hepatoma HepG2 cells through modulation of toll-like receptor negative regulators of mitogen-activated protein kinase and NF-κB signaling. Front Immunol. 9: 1537.ArticlePubMedPMC
- Kawai T, Ikegawa M, Ori D, Akira S. 2024. Decoding toll-like receptors: recent insights and perspectives in innate immunity. Immunity. 57: 649–673. ArticlePubMed
- Kawano M, Miyoshi M, Miyazaki T. 2019. Lactobacillus helveticus SBT2171 induces A20 expression via toll-like receptor 2 signaling and inhibits the lipopolysaccharide-induced activation of nuclear factor-kappa B and mitogen-activated protein kinases in peritoneal macrophages. Front Immunol. 10: 845.ArticlePubMedPMC
- Kim WK, Jang YJ, Han DH, Jeon K, Lee C, et al. 2020a. Lactobacillus paracasei KBL382 administration attenuates atopic dermatitis by modulating immune response and gut microbiota. Gut Microbes. 12: 1819156.Article
- Kim WK, Jang YJ, Seo B, Han DH, Park S, et al. 2019. Administration of Lactobacillus paracasei strains improves immunomodulation and changes the composition of gut microbiota leading to improvement of colitis in mice. J Funct Foods. 52: 565–575. Article
- Kim W, Lee EJ, Bae IH, Myoung K, Kim ST, et al. 2020b. Lactobacillus plantarum-derived extracellular vesicles induce anti-inflammatory M2 macrophage polarization in vitro. J Extracell Vesicles. 9: 1793514.ArticleLink
- Kwon HK, Lee CG, So JS, Chae CS, Hwang JS, et al. 2010. Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc Natl Acad Sci USA. 107: 2159–2164. ArticlePubMedPMC
- Lamubol J, Ohto N, Kuwahara H, Mizuno M. 2021. Lactiplantibacillus plantarum 22A-3-induced TGF-β1 secretion from intestinal epithelial cells stimulates CD103+ DC and Foxp3+ Treg differentiation and ameliorates colitis in mice. Food Funct. 12: 8044–8055. ArticlePubMed
- Lee SW, Liu CW, Hu JY, Chiang LM, Chuu CP, et al. 2017. Suppressors of cytokine signaling in tuberculosis. PLoS One. 12: e0176377. ArticlePubMedPMC
- Li H, Zhang L, Chen L, Zhu Q, Wang W, et al. 2016. Lactobacillus acidophilus alleviates the inflammatory response to enterotoxigenic Escherichia coli K88 via inhibition of the NF-κB and p38 mitogen-activated protein kinase signaling pathways in piglets. BMC Microbiol. 16: 273.ArticlePubMedPMCPDF
- Lin EK, Chang WW, Jhong JH, Tsai WH, Chou CH, et al. 2023. Lacticaseibacillus paracasei GM-080 ameliorates allergic airway inflammation in children with allergic rhinitis: from an animal model to a double-blind, randomized, placebo-controlled trial. Cells. 12: 768.ArticlePubMedPMC
- Liu L, Lu Y, Martinez J, Bi Y, Lian G, et al. 2016. Proinflammatory signal suppresses proliferation and shifts macrophage metabolism from Myc-dependent to HIF1α-dependent. Proc Natl Acad Sci USA. 113: 1564–1569. ArticlePubMedPMC
- Massagué J, Sheppard D. 2023. TGF-β signaling in health and disease. Cell. 186: 4007–4037. ArticlePubMedPMC
- Nakagawa R, Naka T, Tsutsui H, Fujimoto M, Kimura A, et al. 2002. SOCS-1 participates in negative regulation of LPS responses. Immunity. 17: 677–687. ArticlePubMed
- Palsson-McDermott EM, Curtis AM, Goel G, Lauterbach MA, Sheedy FJ, et al. 2015. Pyruvate kinase M2 regulates HIF-1α activity and IL-1β induction and is a critical determinant of the Warburg effect in LPS-activated macrophages. Cell Metab. 21: 65–80. ArticlePubMedPMC
- Pereira M, Gazzinelli RT. 2023. Regulation of innate immune signaling by IRAK proteins. Front Immunol. 14: 1133354.ArticlePubMedPMC
- Priem D, van Loo G, Bertrand MJM. 2020. A20 and cell death-driven inflammation. Trends Immunol. 41: 421–435. ArticlePubMed
- Rahemi S, Nematollahi-Mahani SN, Rajaie A, Fallah H. 2019. Inhibitor of interleukin-1 receptor-associated kinases 1/4, can increase the sensitivity of breast cancer cells to methotrexate. Int J Mol Cell Med. 8: 200–209. ArticlePubMed
- Ren D, Wang D, Liu H, Shen M, Yu H. 2019. Two strains of probiotic Lactobacillus enhance immune response and promote naive T cell polarization to Th1. Food Agric Immunol. 30: 281–295. ArticleLink
- Ryu SH, Park JH, Choi SY, Jeon HY, Park JI, et al. 2016. The probiotic Lactobacillus prevents Citrobacter rodentium-induced murine colitis in a TLR2-dependent manner. J Microbiol Biotechnol. 26: 1333–1340. ArticlePubMed
- Sakai F, Hosoya T, Ono-Ohmachi A, Ukibe K, Ogawa A, et al. 2014. Lactobacillus gasseri SBT2055 induces TGF-β expression in dendritic cells and activates TLR2 signaling to produce IgA in the small intestine. PLoS One. 9: e105370. ArticlePubMedPMC
- Saravanan P, R P, Balachander N, K KRS, S S et al. 2023. Anti-inflammatory and wound healing properties of lactic acid bacteria and its peptides. Folia Microbiol. 68: 337–353. ArticlePDF
- Shen J, Qiao Y, Ran Z, Wang T. 2013. Differential activation of TRAF4 and TRAF6 in inflammatory bowel disease. Mediators Inflamm. 2013: 647936.ArticlePubMedPMC
- Sobah ML, Liongue C, Ward AC. 2021. SOCS proteins in immunity, inflammatory diseases, and immune-related cancer. Front Med. 8: 727987.Article
- Starr T, Bauler TJ, Malik-Kale P, Steele-Mortimer O. 2018. The phorbol 12-myristate-13-acetate differentiation protocol is critical to the interaction of THP-1 macrophages with Salmonella Typhimurium. PLoS One. 13: e0193601. ArticlePubMedPMC
- Sudhakaran AV, Panwar H, Chauhan R, Duary RK, Rathore RK, et al. 2013. Modulation of anti-inflammatory response in lipopolysaccharide-stimulated human THP-1 cell line and mouse model at gene expression level with indigenous putative probiotic lactobacilli. Genes Nutr. 8: 637–648. ArticlePubMedPMCPDF
- Sun KY, Xu DH, Xie C, Plummer S, Tang M, et al. 2017. Lactobacillus paracasei modulates LPS-induced inflammatory cytokine release by monocyte-macrophages via up-regulation of negative regulators of NF-kappaB signaling in a TLR2-dependent manner. Cytokine. 92: 1–11. ArticlePubMed
- Taverniti V, Guglielmetti S. 2011. The immunomodulatory properties of probiotic microorganisms beyond their viability (ghost probiotics: proposal of paraprobiotic concept). Genes Nutr. 6: 261–274. ArticlePubMedPMCPDF
- Thiel FG, Asgarbeik S, Glaubitz J, Wilden A, Lerch MM, et al. 2023. IRAK3-mediated suppression of pro-inflammatory MyD88/IRAK signaling affects disease severity in acute pancreatitis. Sci Rep. 13: 10833.ArticlePubMedPMCPDF
- Wang Z, Wu H, Chen Y, Che H, Wang X, et al. 2021. Lactobacillus paracasei S16 alleviates lumbar disc herniation by modulating inflammatory response and gut microbiota. Front Nutr. 8: 701644.ArticlePubMedPMC
- Wells JM. 2011. Immunomodulatory mechanisms of lactobacilli. Microb Cell Fact. 10: S17.ArticlePubMedPMC
- Xia P, Wu Y, Lian S, Yan L, Meng X, et al. 2021. Research progress on toll-like receptor signal transduction and its roles in antimicrobial immune responses. Appl Microbiol Biotechnol. 105: 5341–5355. ArticlePubMedPMCPDF
- Xiong Y, Qiu F, Piao W, Song C, Wahl LM, et al. 2011. Endotoxin tolerance impairs IL-1 receptor-associated kinase 4 and TGF-β-activated kinase 1 activation, K63-linked polyubiquitination and assembly of IRAK1, TNF receptor-associated factor 6, and IκB kinaseγ and increases A20 expression. J Biol Chem. 286: 7905–7916. ArticlePubMedPMC
- Zarember KA, Godowski PJ. 2002. Tissue expression of human toll-like receptors and differential regulation of toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J Immunol. 168: 554–561. ArticlePubMedPDF
- Zeng Z, Guo X, Zhang J, Yuan Q, Chen S. 2021. Lactobacillus paracasei modulates the gut microbiota and improves inflammation in type 2 diabetic rats. Food Funct. 12: 6809–6820. ArticlePubMed
References
Figure & Data
References
Citations

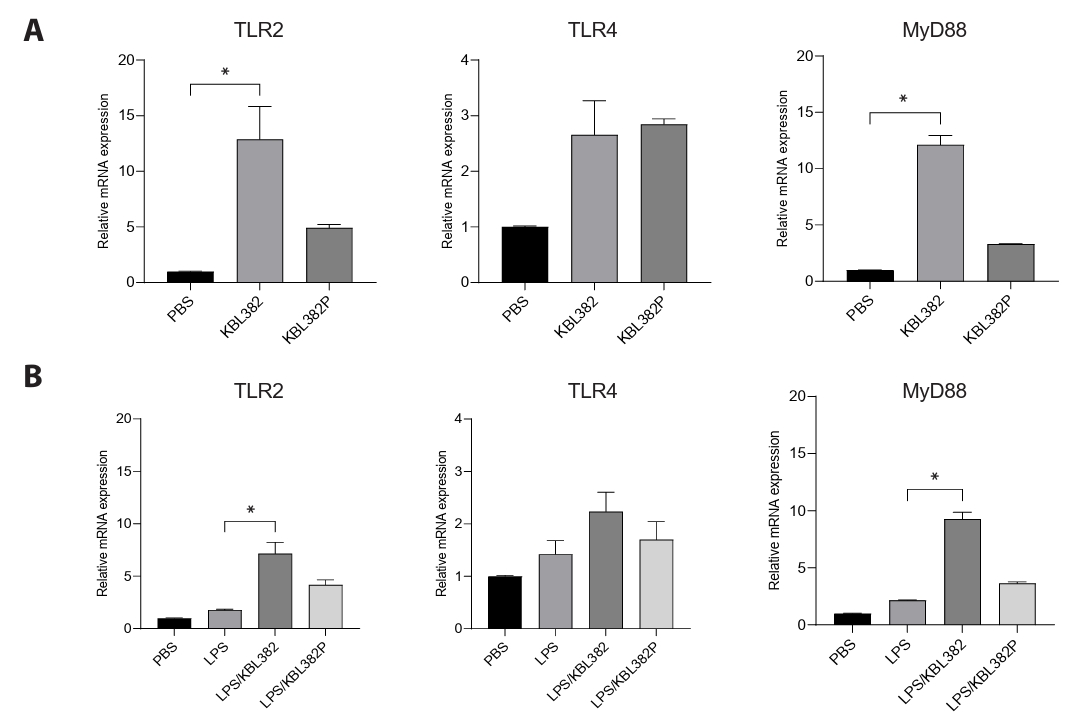
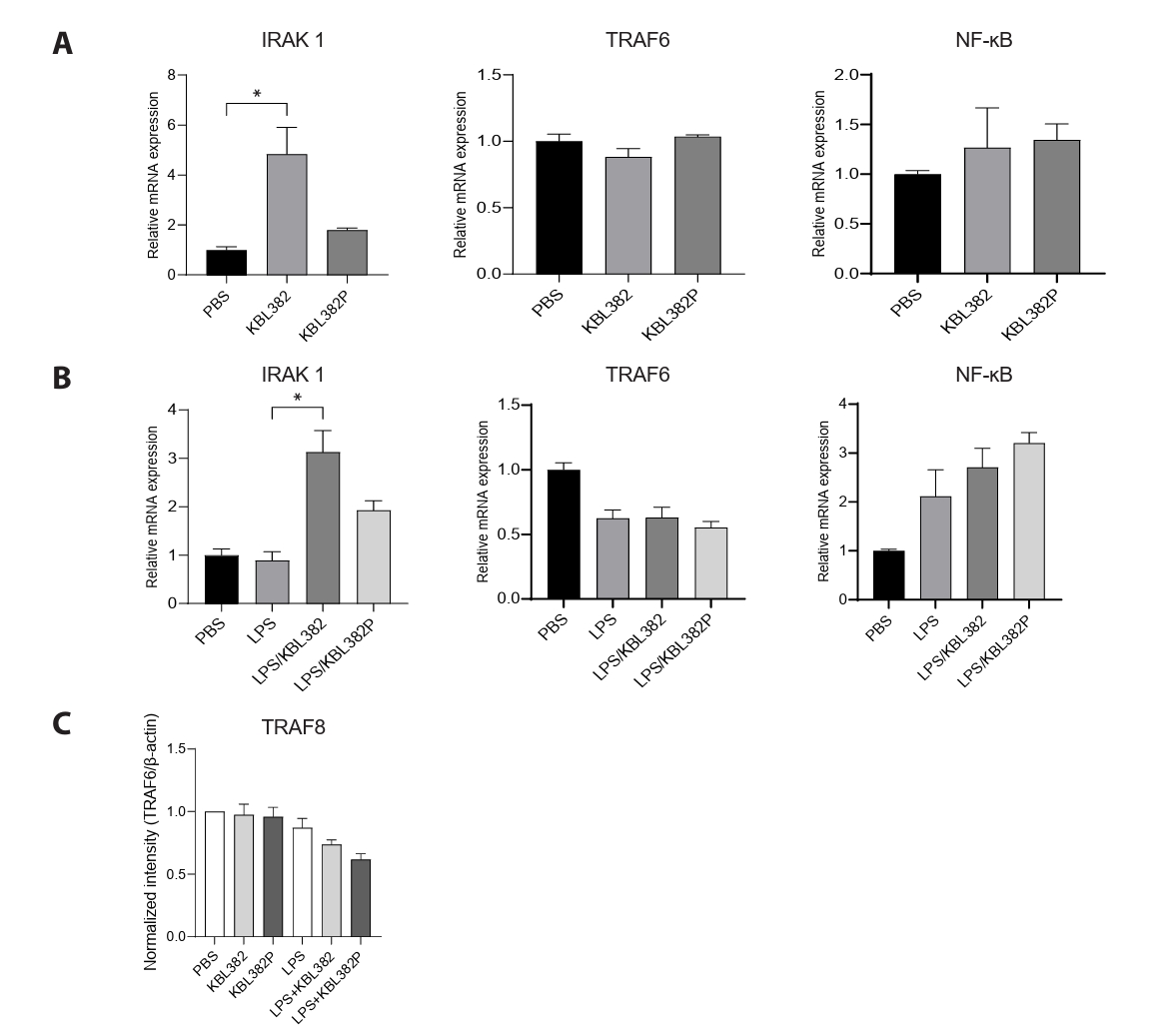
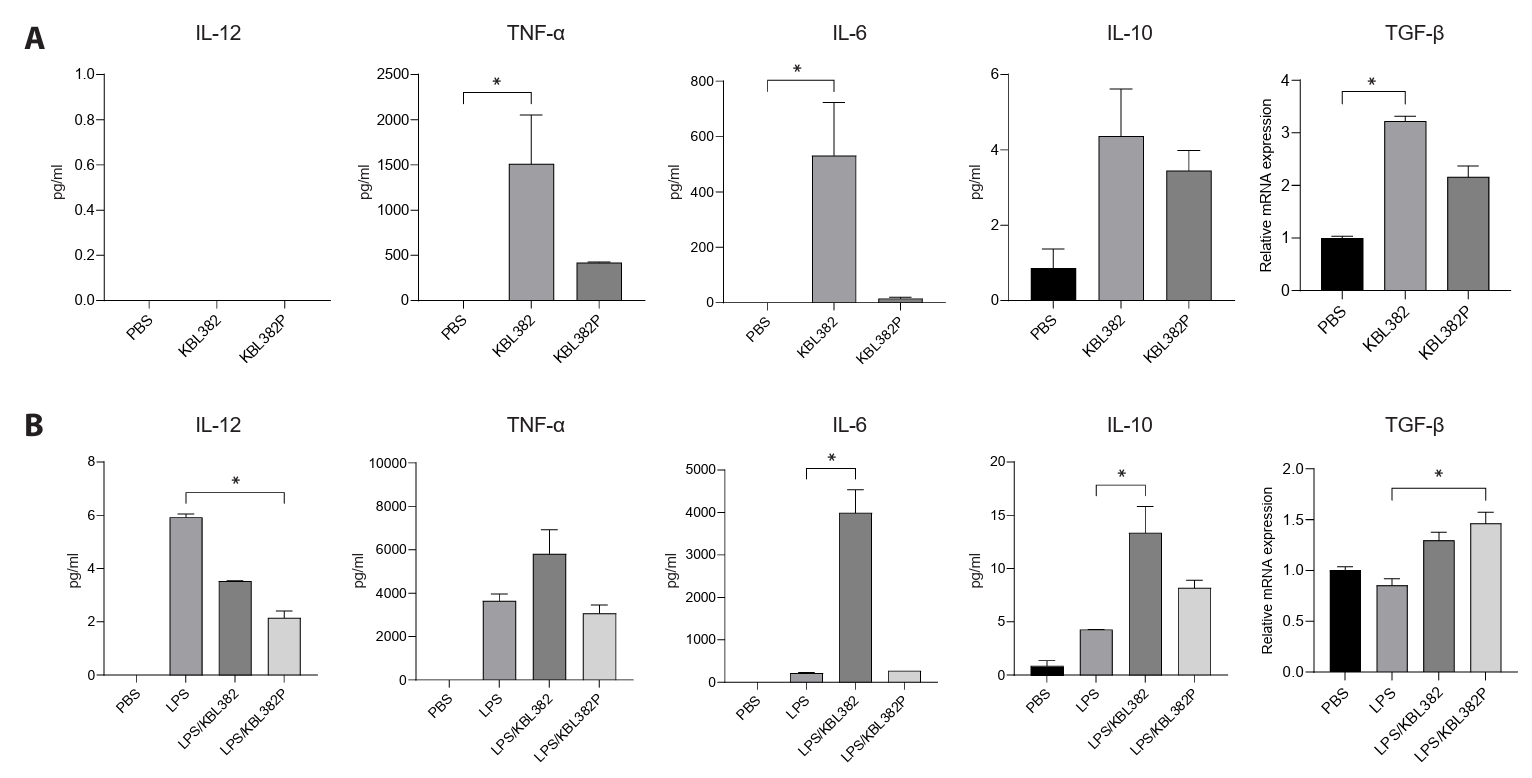
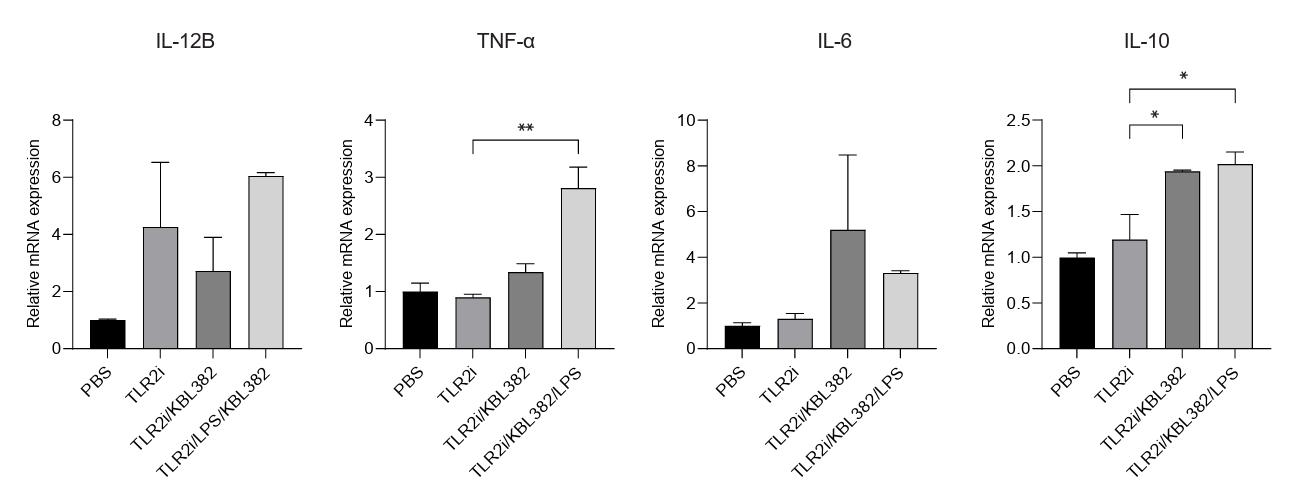
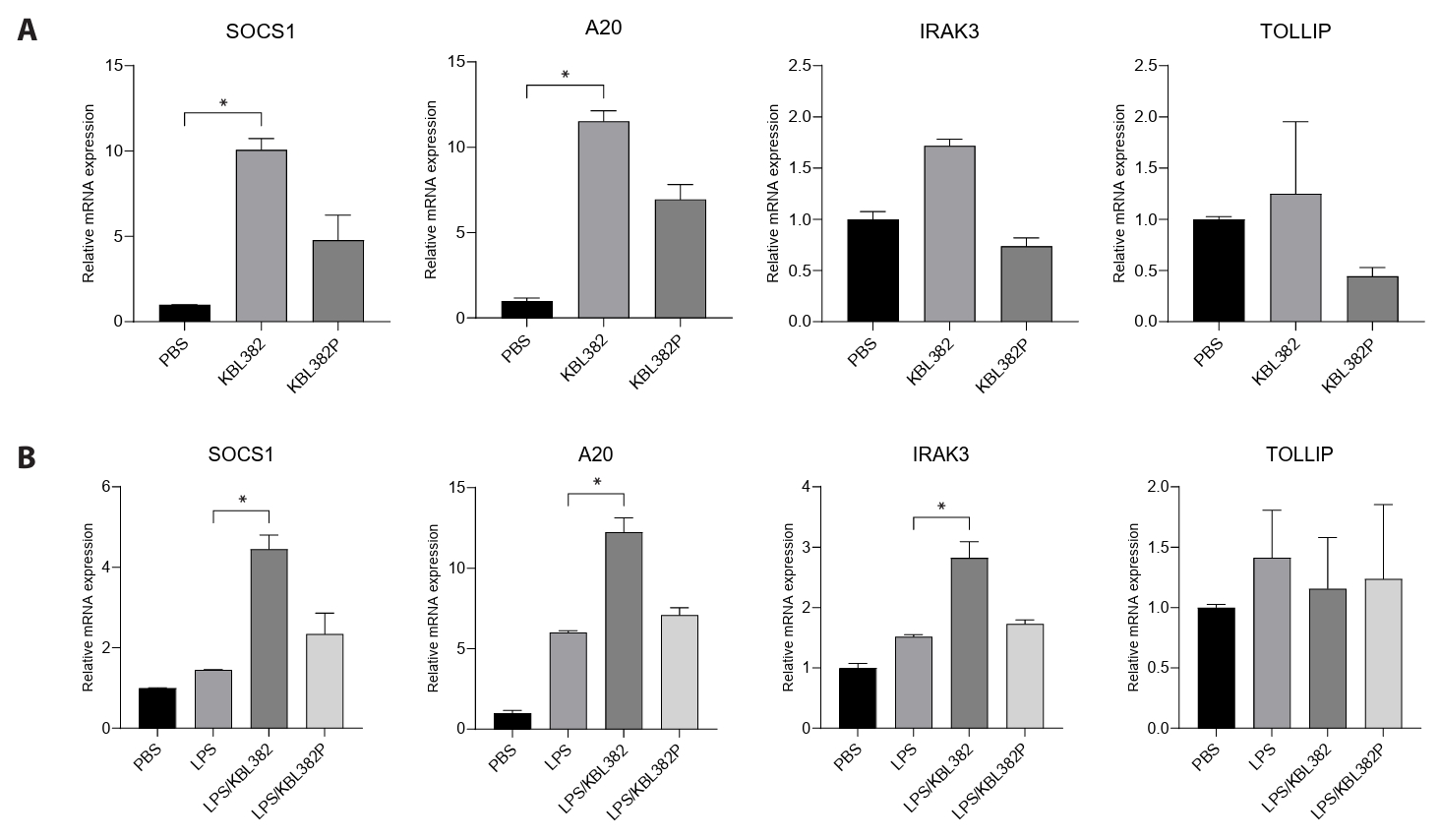
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
| Target | Sequence | Reference |
|---|---|---|
| A20 | Fw: 5’- AACATTTTGCTGCTGCCTC -3’ | |
| Rv: 5’- AGGTGCTTTGTGTGGTTCG -3’ | ||
| GAPDH | Fw: 5’- GGAAGGTGAAGGTCGGAGTC -3’ | |
| Rv: 5’- TCAGCCTTGACGGTGCCATG -3’ | ||
| IL-6 | Fw: 5’- CATCCTCGACGGCATCTCAG -3’ | |
| Rv: 5’- GCTCTGTTGCCTGGTCCTC -3’ | ||
| IL-10 | Fw: 5’- TCAGGGTGGCGACTCTAT -3’ | |
| Rv: 5’- TGGGCTTCTTCTAAATCGTTC -3’ | ||
| IL-12B | Fw: 5’- GGCTCCATGAAGGTGCTAC -3’ | |
| Rv: 5’- GTTCAGCCTCAGAATGCAAAA -3’ | ||
| IRAK1 | Fw: 5’- ACTGGCCCTTGGCAGCTC -3’ | |
| Rv: 5’- GGCCAGCTTCTGGACCATC -3’ | ||
| IRAK3 | Fw: 5’- TGCAACGCGGGCAAA -3’ | |
| Rv: 5’- TTTAGTGATGTGGGAGGATCTTCA -3’ | ||
| MyD88 | Fw: 5’- GAGCGTTTCGATGCTTCAT -3’ | |
| Rv: 5’- CGGATCATCTCCTGCACAAA -3’ | ||
| NF-κB | Fw: 5’- TCAATGGCTACACAGGACCA -3’ | |
| Rv: 5’- CACTGTCACCTGGAAGCAGA -3’ | ||
| SOCS1 | Fw: 5’- CTGGGATGCCGTGTTATTTT-3’ | |
| Rv: 5’- TAGGAGGTGCGAGTTCAGGT-3’ | ||
| TLR2 | Fw: 5’- GCCAAAGTCTTGATTGATTGG-3’ | |
| Rv: 5’- TTGAAGTTCTCCAGCTCCTGG -3’ | ||
| TLR4 | Fw: 5’- GGTGGAAGTTGAACGAATGG -3’ | |
| Rv: 5’- CCAGCAAGAAGCATCAGGTG -3’ | ||
| TNF-α | Fw: 5’- TCTCGAACCCCGAGTGACAA -3’ | |
| Rv: 5’- TATCTCTCAGCTCCACGCCA -3’ | ||
| TOLLIP | Fw: 5’-AGGTGACAACTGTCTCCGTC-3’ | |
| Rv: 5’-GCCAACTTTGCCTGTACCAC-3’ | ||
| TRAF6 | Fw: 5’-CCTTTGGCAAATGTCATCTGTG-3’ | |
| Rv: 5’-CTCTGCATCTTTTCATGGCAAC-3’ | ||
| TGF-β | Fw: 5′-GAA GGC AGA GTT CAG GGT CTT-3′ | |
| Rv: 5′-GGT TCC TGT CTT TGT GGT GAA-3′ |
Fw: Represents a forward primer sequence. Rv: Represents a reverse primer sequence.
Table 1.
TOP
 MSK
MSK
 Cite this Article
Cite this Article






