- About
- Browse Articles
-
Special Issues
- Pioneering strategies for overcoming bacterial drug resistance (2026)
- Advancing microbial engineering through synthetic biology (2025)
- Host-associated microbiome (2024)
- Bacterial regulatory mechanisms for the control of complex cellular mechanisms (2023)
- Two years into COVID-19 pandemic: Where are we? (2022)
- Collections
- For Contributors
- Policies
- E-Submission

- About
- Browse Articles
-
Special Issues
- Pioneering strategies for overcoming bacterial drug resistance (2026)
- Advancing microbial engineering through synthetic biology (2025)
- Host-associated microbiome (2024)
- Bacterial regulatory mechanisms for the control of complex cellular mechanisms (2023)
- Two years into COVID-19 pandemic: Where are we? (2022)
- Collections
- Policies
- For Contributors
Articles
- Page Path
- HOME > J. Microbiol > Volume 63(3); 2025 > Review
-
Review
Progress and challenges in CRISPR/Cas applications in microalgae - Quynh-Giao Tran1,†, Trang Thi Le1,2,†, Dong-Yun Choi1, Dae-Hyun Cho1, Jin-Ho Yun1,2, Hong Il Choi1,2, Hee-Sik Kim1,2,*, Yong Jae Lee1,2,*
-
Journal of Microbiology 2025;63(3):e2501028.
DOI: https://doi.org/10.71150/jm.2501028
Published online: March 28, 2025
1Cell Factory Research Center, Korea Research Institute of Bioscience and Biotechnology (KRIBB), Daejeon 34141, Republic of Korea
2Department of Environmental Biotechnology, KRIBB School of Biotechnology, University of Science and Technology (UST), Daejeon 34113, Republic of Korea
- *Correspondence Hee-Sik Kim hkim@kribb.re.kr Yong Jae Lee leeyj@kribb.re.kr
- †These authors contributed equally to this work.
© The Microbiological Society of Korea
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) technologies have emerged as powerful tools for precise genome editing, leading to a revolution in genetic research and biotechnology across diverse organisms including microalgae. Since the 1950s, microalgal production has evolved from initial cultivation under controlled conditions to advanced metabolic engineering to meet industrial demands. However, effective genetic modification in microalgae has faced significant challenges, including issues with transformation efficiency, limited target selection, and genetic differences between species, as interspecies genetic variation limits the use of genetic tools from one species to another. This review summarized recent advancements in CRISPR systems applied to microalgae, with a focus on improving gene editing precision and efficiency, while addressing organism-specific challenges. We also discuss notable successes in utilizing the class 2 CRISPR-associated (Cas) proteins, including Cas9 and Cas12a, as well as emerging CRISPR-based approaches tailored to overcome microalgal cellular barriers. Additionally, we propose future perspectives for utilizing CRISPR/Cas strategies in microalgal biotechnology.
Introduction
Milestones in the Application of CRISPR/Cas System in Microalgae
CRISPR Toolboxes for Microalgal Gene Editing
Challenges and Solutions in Utilizing CRISPR/Cas System in Microalgae
Applications of CRISPR/Cas in Microalgae
Perspectives and Future Directions
Conclusion
Acknowledgments
This work was supported by the Basic Science Research Program (2021R1C1C100342; Y.J.L.) and the STEAM Project (RS-2024-00459155; H.S.K.) of the National Research Foundation (NRF) funded by the Korean government (MSIT).
Author Contributions
Y.J.L. and H.S.K. were responsible for funding acquisition, supervising the project, and reviewing the manuscript. Q.G.T. contributed to the conceptualization and writing of the manuscript. T.T.L. contributed to data collection and writing. J.H.Y., H.I.C., D.Y.C., and D.H.C. provided feedback on the writing process. All authors reviewed and proofread the final version of the article. †Q.G.T. and †T.T.L. contributed equally to this work.
Conflict of Interest
The authors declare that there is no conflict of interest.
| Algal strain | CRISPR/Cas system |
Strategy |
Targeted genes | Selection Marker | Products | Mutagenesis efficiency (%)⸶ | Targeted mutagenesis frequency⸷ | References | |
|---|---|---|---|---|---|---|---|---|---|
| Approaches | Delivery method | ||||||||
| Chlamydomonas reinhardtii | Cas9 | Vector driven | Electroporation | FKB12 | Rapamycin | First successful transient expression of Cas9 and sgRNA genes in C. reinhardtii | 0.0014 | 2 × 10-8 | Jiang et al. (2014) |
| Cas9 | RNP | Electroporation | MAA7, CpSRP43, ChlM | Auxotrophic selection using 5-FI | Visible auxotrophic colonies with mutations targeted at the Cas9 cut sites | 40% (MAA7), 1.4% (CpSRP43), and 0.17% (ChlM) | 8.9 × 10-8 (MAA7), 3.3 × 10-8 (CpSRP43), and 5 × 10-8 (ChlM) | Shin et al. (2016) | |
| Cas9 | RNP | Electroporation | CpFTSY, ZEP | Based on the coloration of the cells | High zeaxanthin-producing mutants with improved photosynthetic productivity | 0.46 to 0.56% | Not mention | Baek et al. (2016) | |
| CRISPRi | Vector driven | Glass beads | PEPC1 | Hygromycin and paromomycin | A 74.4% increase in lipid content and a 94.2% enhancement in lipid productivity | 0.94 | Not mention | Kao & Ng (2017) | |
| Cas9 | Vector driven/RNP | Electroporation | COP1/2, COP3, COP4, COP5, PHOT, UVR8, VGCC, MAT3, aCRY, PSY1 | Paromomycin | Protocols for the rapid isolation of non-selectable gene mutants | 5 to 15% | 2.5 × 10˗5 to 2.5 × 10-4 | Greiner et al. (2017) | |
| Cas9 | Vector driven/RNP | Electroporation | FKB12, ALS, ARG | Rapamycin and zeocin | Gene-within-a-gene hybrid construct, composed of a Cas9 gene containing an artificial intron and an inserted sgRNA gene | Not mention | 3 × 10-8 | Jiang & Weeks (2017) | |
| Cpf1 (Cas12a) | RNP | Electroporation | FKB12, CpFTSY, CpSRP43, PHT7 | Rapamycin and based on coloration and chlorophyll fluorescence | CRISPR/Cpf1-mediated DNA editing efficiencies increased 500-fold with the use of single-stranded oligodeoxynucleotides (ssODNs) | ∼10% | Not mention | Ferenczi et al. (2017) | |
| Cas9 | RNP | Electroporation | PLA2 | Hygromycin | Lipid productivity in phospholipase A2 knockout mutants increased by 64.25%, reaching 80.92 g/L/d | Not mention | Not mention | Shin et al. (2019) | |
| Cas9 | RNP | Electroporation | ELT1 | Hygromycin | Total lipid accumulated up to 28% of dry biomass, with a 27.2% increase in C18:1 ratio | 0.1368 | Not mention | Nguyen et al. (2020) | |
| Cas9 | RNP | Electroporation | LCYE, ZEP | Hygromycin | Mutant with 60% higher zeaxanthin yield (5.24 mg/L) and content (7.28 mg/g) | Not mention | Not mention | Song et al. (2020) | |
| Cas9 | RNP | Electroporation and glass beads | IFT81, FAP70, MOT17, CDPK13, CEP131 | Paromomycin | Development of CRISPR-based targeted insertional mutagenesis method (TIM) for C. reinhardtii | 40% to 95% | Not mention | Picariello et al. (2020) | |
| Cas9 | RNP | Cell-penetrating peptide pVEC | Maa7, FKB12 | Auxotrophic selection using 5-FI (MAA7) and Rapamycin (FKB12) | Delivery of Cas9/sgRNA RNP into C. reinhardtii using cell-penetrating peptide pVEC | 8.41% to 46.56% | Not mention | Kang et al. (2020) | |
| Cas9 | RNP | Electroporation | PPX1, FTSY, WDTC1 | Paromomycin or oxyfluorfen | Generation of individual strains with precise mutations in multiple target genes | 1.3% (PPX1 and FTSY) and 0.8% (WDTC1) | Not mention | Akella et al. (2021) | |
| Cas9 | RNP | Electroporation | SPD1 | Paromomycin or hygromycin | Targeted knockout of CrSPD1 induces spermidine auxotroph, which could be used as a selectable marker in biotechnology | 10% to 66% | Not mention | Freudenberg et al. (2022) | |
| Cas9 | RNP | Glass bead | NR | Hygromycin | Generation of mutants with a bacterial phytase gene cassette knocking into the NR gene | 0.1481 | Not mention | Zadabbas Shahabadi et al. (2023) | |
| Cas9 | RNP | Electroporation | PSR1, CpFTSY | 30% starch | Generation of mutants with impaired extracellular phosphatase synthesis in response to Pi deprivation | 0.22 | Not mention | Battarra et al. (2024) | |
| Cas9 | RNP | Electroporation | LCYE | Hygromycin | A 2.3-fold increase in astaxanthin accumulation in the ΔLCYE mutant | 0.0417 | Not mention | Kneip et al. (2024) | |
| Nannochloropsis spp. | Cas9 | Vector driven | Electroporation | NR | Grow normally under NH4Cl but fail to grow under NaNO3 | Knockout of the NR gene | 0.1% to 1% | Not mention | Wang et al. (2016) |
| Cas9 | In vitro–synthesized gRNAs/Cas9-expressing line | Electroporation | Insertional mutagenesis of 18 gene of 20 transcription factors | Blasticidin or Hygromycin | Improvement in the total carbon-to-lipid ratio from 20% (wild type) to 40–55% (mutants) | 6.25 to 78% | Not mention | Ajjawi et al. (2017) | |
| Cas9 | In vitro–synthesized gRNAs/reporter-free Cas9-expressing line | Electroporation | Aco1 | Blasticidin and Hygromycin | Mutants with doubled lipid productivity and ∼50% reduced photosynthetic antenna size | ∼50% | Not mention | Verruto et al. (2018) | |
| Cas9 and Cas12a | RNP | Electroporation | NR | Zeocin | FnCas12a generated HDR-based mutants with up to 93% efficiency | 34–71% (for Cas9) and 3-93% (for Cas12a) | Not mention | Naduthodi et al. (2019) | |
| Cas9 | In vitro–synthesized gRNAs/reporter-free Cas9- expressing line | Electroporation | Knock-in of FAD12 gene at the T1 hotspot | Zeocin | Improved production of polyunsaturated fatty acids (PUFAs) | 0.714 | Not mention | Ryu et al. (2021) | |
| Cas12a | RNP | Electroporation | NR | Grow normally under NH4Cl but fail to grow under NaNO3 | Generation of markerless genome editing tool to knock out target genes | 0.1052 | Not mention | Naduthodi et al. (2021) | |
| Cas9 | Vector driven | Electroporation | LER1 and LER2 | Hygromycin | Double deletion of both LER1 and LER2 (from chromosome 9), total ~214 kb | 0.285 | Not mention | Wang et al. (2021) | |
| dCas9 | Vector driven | Electroporation | g1248 | Zeocin | Growth and photosynthetic parameters (Fv/Fm) of the mutants increased by 23% and 12%, respectively, compared to the wild type under ambient CO2 levels | Not mention | Not mention | Wei et al. (2022) | |
| Cas9 | Vector driven | Bombardment | LSMT | Hygromycin | Mutation induced 18–20% reduction in fructose-1,6-bisphosphate aldolases, along with 9.7–13.8% increase in dry weight and enhanced growth | 0.1 | 2 × 10-8 | Liang et al. (2024) | |
| Phaeodactylum tricornutum | Cas9 | Vector driven | Bombardment | CpSRP54 | Zeocin | Generation of stable targeted gene mutations in marine algae | 0.31 | Not mention | Nymark et al. (2016) |
| Cas9 | Vector-driven and RNP | Bombardment | PtUMPS, PtAPT, PtAureo1a | Nourseothricin, 2-FA, adenine, and uracil | A single-step generation of triple knockout strains | 65% to 100% | Not mention | Serif et al. (2018) | |
| Cas9 | Vector driven | Bacterial conjugation | Phatr3_J46193 | Phleomycin | Bacterial conjugation-mediated Cas9 delivery to minimize genome exposure to nuclease activity | Not mention | 2 × 10-5 | Russo et al. (2018) | |
| Cas9 | Vector driven | Electroporation | LACS | Zeocin | Reduced growth rate and altered molecular profiles of PC and TAGs | Not mention | Not mention | Hao et al. (2022) | |
| Cas9 | Vector driven | Conjugation of plasmids | CryP | Zeocin | Increased light-harvesting protein levels in CryP knockout mutants | Not mention | Not mention | Yang et al. (2022) | |
| Cas9 | Vector driven | Bombardment | PtTHIC and PtSSSP | Zeocin | Targeted mutation of the TPP aptamer in the THIC gene encoding HMP-P synthase does not affect thiamine biosynthesis in P. tricornutum | Not mention | Not mention | Llavero-Pasquina et al. (2022) | |
| Cas9 | Vector driven | Bombardment | CpFTSY | Zeocin | Generation and characterization of CpFTSY mutants | Not mention | Not mention | Nymark et al. (2023) | |
| Cas9 | Vector driven | Bombardment | FucT | Zeocin | Knockout of PtFucT1 affected PtGnTI activity in the complex, converting the N-glycan to a mannose-type N-glycan | 0.333 | Not mention | Xie et al. (2023) | |
| Cas9 | Vector driven | Electroporation | StLDP | Zeocin | StLDP functions as an LD scaffold protein in P. tricornutum, regulating LD numbers in the stldp mutant and complemented strains | 0.8125 | Not mention | Yoneda et al. (2023) | |
| Cas9 | Vector driven | Bacterial conjugation | ZEP 2, ZEP3 | No selective pressure | Generation of zep mutants as a platform for diatoxanthin production | Not mention | Not mention | Graesholt et al. (2024) | |
| Chlorella sp. | Cas9 | Vector driven | Electroporation | fad3 | Hygromycin | Mutants have 46% higher lipid accumulation | Not mention | Not mention | Lin & Ng (2020) |
| Cas9 | Vector driven and RNP | Bombardment | NR, APT | NaNO2 and KClO3 | Generation of auxotrophic strains | Not mention | Not mention | Kim et al. (2021) | |
| CRISPRi and CRISPRa | Vector driven | Electroporation | Randomly mediate gene regulation | Hygromycin | Mutants with protein content of 60% to 65% (w/w) of dry cell weight | 0.5 | Not mention | Lin et al. (2022) | |
| Cas9 | Vector driven | Electroporation | GS | Hygromycin | Generation of mutants with enhanced biomass, protein, and lutein content | Not mention | Not mention | Teng & Ng (2023) | |
| Cas9 | Vector driven | Electroporation | APT | Hygromycin | Combination of Alcalase treatment with PEG transformation for efficient gene editing in Chlorella | Not mention | 1 × 10-7 to 2 × 10-7 | Kim et al. (2024) | |
| Porphyridium purpureum | Cas9 | RNP | Bombardment | CHS1 | No selective pressure | Generation of chlorophyll synthase loss-of-function mutants with increased phycoerythrin levels | Not mention | Not mention | Jeon et al. (2021) |
| Tetraselmis sp. | Cas9 | RNP | Bombardment | AGP | No selective pressure | Mutants had 2.7- and 3.1-fold increased lipid content (21.1% and 24.1% of DCW, respectively) | Not mention | Chang et al. (2020) | |
| Euglena gracilis | Cas9 | RNP | Electroporation | EgGSL2 | No selective pressure | Transgene-free targeted mutagenesis and ssODN-mediated gene knockin | 77.7 to 90.1% | Not mention | Nomura et al. (2019) |
| LbCas12a | RNP | Electroporation | EgGSL2, EgcrtB | No selective pressure | High-efficiency genome editing system using direct delivery of LbCas12a RNP complexes | 77.2–94.5% | Not mention | Nomura et al. (2024) | |
| Cas9 | RNP | Electroporation | Knockout of 16 carotenoid biosynthetic genes present in E. gracilis | No selective pressure | Mutants with different carotenoid compositions | Not mention | Not mention | Tamaki et al. (2023) | |
| Thalassiosira pseudonana | Cas9 | Vector driven | Bombardment | Urease | Nourseothricin | Significant reduction in growth rate and cell size compared to nitrate growth | 0.121 | 8 × 10-7 | Hopes et al. (2016) |
| Cas9 | Vector driven | Bombardment | Sin1 | Nourseothricin | Mutants exhibit reduced biosilica content and morphological aberrations, affecting cell wall strength and stiffness | Not mention | Not mention | Görlich et al. (2019) | |
| Cas9 nickase | Vector driven | Bombardment | TpθCA3 | Nourseothricin | Development of an efficient Cas9 nickase (D10A) system for highly specific indel introduction into target DNA | 0.39 | 3.4 × 10-7 | Nawaly et al. (2020) | |
⸶Mutagenesis efficiency (%) was determined by calculating the proportion of mutants confirmed through genotyping methods, such as Sanger sequencing or polymerase chain reaction (PCR), against the total number of mutants subjected to genotyping.
⸷Targeted mutagenesis efficiency was defined by the number of mutants confirmed by genotyping in relation to the initial size of the mutant pool.
Aco1, acyl-CoA oxidase; AGP,ADP-glucose pyrophosphorylase; ALS, acetolactate synthase; ARG, argininosuccinate lyase; APT, adenine phosphoribosyl transferase; CDPK13, calcium-dependent protein kinase 13; ChlM, Mg-protoporphyrin IX S-adenosyl methionine O-methyl transferase; CHS1, chlorophyll synthase; CpFTSY, chloroplast signal recognition particle receptor protein; CpSRP, chloroplast signal recognition particle; CpSRP43, chloroplast SRP43; CryP, cryptochrome; ELT1, esterase/lipase/thioesterase 1; EgGSL2, glucan synthase-like 2; EgcrtB, phytoene synthase gene; FAD12, Δ12-fatty acid desaturases; FAP 70, flagella-associated proteins 70; FucT, fucosyltransferase; fad3, omega-3 fatty acid desaturase; GS, glutamate synthase; IFT, Intraflagellar transport; LACS, Long-chain acyl-CoA synthetases; LER, low expression region; LCYE, lycopene epsilon cyclase; LSMT, rubisco large-subunit methyltransferase; MAA7, beta-subunit of tryptophan synthase; NR, nitrate reductase; PEPC1, phosphoenolpyruvate carboxylase 1; PLA2, phospholipase A2; PPX1, protoporphyrinogen IX oxidase; PSY, phytoene synthase-1; SPD1, spermidine synthase gene; Sin1, silicanin-1; StLDP, stramenopile-type LD protein; TpθCA3, θ-type carbonic anhydrase; VGCC, voltage-gated calcium channel; ZEP, zeaxanthin epoxidase; Phatr3_J46193, P. tricornutum chr9: 533409–537647 locus; g1248: potential methyltransferase responsible for DNA or mRNA methylation.
| Components | Source/available toolkits | Company |
|---|---|---|
| Plasmids | www.addgene.org | Addgene (USA) |
| www.snapgene.com | SnapGene Plasmid Database (USA) | |
| www.genscript.com | GenScript (USA) | |
| www.idtdna.com | IDT (USA) | |
| Cas9 | http://www.toolgen.com/ko | ToolGen, Inc. (Korea) |
| https://www.macrogen.com/ko/main | Macrogen, Inc. (Korea) | |
| https://sg.idtdna.com/page | IDT (USA) | |
| https://www.neb.com/en | NEB (USA) | |
| https://www.takara.co.kr/ | TaKaRa (Japan) | |
| gRNA | MEGAshortscriptTM T7 Kit | Ambion (USA) |
| HiScribe T7 RNA Kit | NEB (USA) | |
| CUGA7 gRNA Synthesis Kit | Nippon Gene (Japan) | |
| EnGen® sgRNA Synthesis Kit | NEB (USA) | |
| Guide-itTM sgRNA In Vitro Transcription | TaKaRa (Japan) |
| Microalgal strains | Variants of Cas protein | Origin | Codon optimization | PAM | References |
|---|---|---|---|---|---|
| C. reinhardtii | SpCas9 | Streptococcus pyogenes | Yes | NGG | Jiang et al. (2014) |
| C. reinhardtii | SaCas9 and SpCas9 | Staphylococcus aureus and S. pyogenes | Yes | NGG | Greiner et al. (2017), Lee et al. (2022) |
| C. reinhardtii | SpCas9 | S. pyogenes | Yes | NGG | Jiang & Weeks (2017) |
| C. reinhardtii | Dead SpCas9(dCas9) | S. pyogenes | Maize codon–optimized | NGG | Kao & Ng (2017) |
| P. tricornutum | SpCas9 | S. pyogenes | Yes | NGG | Graesholt et al. (2024), Nymark et al. (2023), Nymark et al. (2016), Russo et al. (2018), Serif et al. (2018), Yoneda et al. (2023) |
| N. oceanica IMET1 | SpCas9 | S. pyogenes | Yes | NGG | Wang et al. (2016) |
| N. gaditana | SpCas9 | S. pyogenes | Yes | NGG | Ajjawi et al. (2017), Verruto et al. (2018) |
| N. salina | SpCas9 | S. pyogenes | Chlamydomonas-codon optimized | NGG | Ryu et al. (2021) |
| N. oceanica | dCas9 | S. pyogenes | Yes | NGG | Wei et al. (2022) |
| T. pseudonana | SpCas9 | S. pyogenes | Human-codon optimized | NGG | Görlich et al. (2019), Hopes et al. (2016) |
| T. pseudonana | SpCas9 | S. pyogenes | Yes | NGG | Nawaly et al. (2020) |
| C. vulgaris | SpCas9 | S. pyogenes | Yes | NGG | Kim et al. (2021) |
| C. vulgaris | SpCas9 | S. pyogenes | Maize-codon optimized | NGG | Lin & Ng (2020) |
| C. sorokiniana | dCas9 | S. pyogenes | Maize-codon optimized | NGG | Lin et al. (2022) |
| Features | Microalgal strains | Host | References | |
|---|---|---|---|---|
| Cas9 | pCaMV 35S/tNOS | C. reinhardtii | Cauliflower mosaic virus (CaMV) | Jiang et al. (2014), Jiang & Weeks (2017), Kao & Ng (2017) |
| C. vulgaris | Kim et al. (2021), Lin & Ng (2020) | |||
| C. sorokiniana | Kim et al. (2024), Lin et al. (2022) | |||
| pHSP70A-RBCS2/RBCS2 3’UTR | C. reinhardtii | C. reinhardtii | Greiner et al. (2017) | |
| pPsaD/tPsaD | C. reinhardtii | C. reinhardtii | Jiang & Weeks (2017) | |
| pLHCF2/tLHCF1 | P. tricornutum | P. tricornutum | Nymark et al. (2023), Nymark et al. (2016), Russo et al. (2018) | |
| pVCP /tATUB | N. oceanica IMET1 | N. oceanica IMET1 | Wang et al. (2016) | |
| pRPL24/ tFRD | N. gaditana | N. gaditana | Ajjawi et al. (2017), Verruto et al. (2018) | |
| pRibi/tldsp | N. oceanica | N. oceanica | Liang et al. (2024), Wang et al. (2021), Wei et al. (2022) | |
| pTpFCP/tNAT | T. pseudonana | T. pseudonana | Hopes et al. (2016) | |
| pNR (nitrate reductase promoter)/tNR (nitrate reductase terminator) | T. pseudonana | T. pseudonana | Chang et al. (2020), Görlich et al. (2019), Nawaly et al. (2020) | |
| pFcpB/tFcpA | P. tricornutum | P. tricornutum | Yang et al. (2022) | |
| sgRNA | pU6/T6 | C. reinhardtii | Arabidopsis | Jiang et al. (2014), Jiang & Weeks (2017), Kao & Ng (2017) |
| C. reinhardtii | C. reinhardtii | Greiner et al. (2017), Lee et al. (2022) | ||
| P. tricornutum | P. tricornutum | Llavero-Pasquina et al. (2022), Nymark et al. (2023), Nymark et al. (2016), Russo et al. (2018), Serif et al. (2018), Yang et al. (2022) | ||
| T. pseudonana | T. pseudonana | Chang et al. (2020), Görlich et al. (2019), Hopes et al. (2016), Nawaly et al. (2020), Nymark et al. (2016), Yang et al. (2022) | ||
| C. vulgaris | Arabidopsis | Kim et al. (2021), Lin & Ng (2020) | ||
| C. sorokiniana | Arabidopsis | Kim et al. (2024), Lin et al. (2022) | ||
| pATPase/tfd | N. oceanica IMET1 | N. oceanica IMET1 | Wang et al. (2016) | |
| pPsaD/tPsaD | C. reinhardtii | C. reinhardtii | Jiang & Weeks (2017) | |
| pRibi/tcs | N. oceanica | N. oceanica | Liang et al. (2024), Wang et al. (2021), Wei et al. (2022) | |
| Donor DNA | pβ-tub/trbcS2 (for HygR) | C. reinhardtii | C. reinhardtii | Nguyen et al. (2020), Shin et al. (2019) |
| pVCP/tVCP (for Sh bleR) | N. oceanica IMET1 | N. oceanica IMET1 | Naduthodi et al. (2019) | |
| pβ-tub/tCOP1 (for aphVII) | C. reinhardtii | C. reinhardtii | Picariello et al. (2020) | |
| pHSP70A/RBCS2 3’UTR (for bacterial phytase gene) | C. reinhardtii | C. reinhardtii | Zadabbas Shahabadi et al. (2023) | |
| pTUB2/tCOP21 (for aphVIII) | C. reinhardtii | C. reinhardtii | Kneip et al. (2024) | |
| Selection marker | pCaMV 35S/tNOS (for HygR) | C. reinhardtii | CaMV | Jiang et al. (2014), Kao & Ng (2017) |
| pFCP/pFCP (for Sh bleR) | P. tricornutum | P. tricornutum | Nymark et al. (2016) | |
| pHSP70-RBCS2/RBCS2-3UTR (for aphVIII) | C. reinhardtii | C. reinhardtii | Kao & Ng (2017) | |
| pPsaD/tPsaD (for Sh bleR) | C. reinhardtii | C. reinhardtii | Jiang & Weeks (2017) | |
| pTCT/ tEIF3 (for blasticidin deaminase) | N. gaditana | N. gaditana | Ajjawi et al. (2017) | |
| pEIF3/tFRD (for HygR) | N. gaditana | N. gaditana | Ajjawi et al. (2017) | |
| pUEP/tUEP (for Sh bleR) | N. salina | N. salina | Ryu et al. (2021) |
Promoter and terminator abbreviations: pCaMV 35S/tNOS, Cauliflower Mosaic Virus 35S promoter/nopaline synthase terminator; pHSP70A-RBCS2/RBCS2 3’UTR, heat shock protein 70A/ribulose-1,5-bisphosphate carboxylase small subunit 2 tandem chimeric promoter /3' untranslated region of ribulose-1,5-bisphosphate carboxylase small subunit 2; pLHCF2/tLHCF1, pVCP/tATUB, violaxanthin/chlorophyll a binding protein promotor/α-tubulin terminator; pRibi/tldsp, ribosomal subunit bidirectional promoter/lipid droplet surface protein terminator; pNR/tNR, nitrate reductase promoter/ nitrate reductase terminator; pATPase/tfd, V-type ATPase promotor/ferredoxin terminator; pRibi/tcs, ribosomal subunit bidirectional promoter/cellulose synthase terminator; pβ-tub/trbcS2, β-tubulin promoter/ribulose-1,5-bisphosphate carboxylase small subunit 2 terminator; pFCP/pFCP, fucoxanthin, chlorophyll a/c-binding protein gene promoter/terminator; pUEP/tUEP, ubiquitin extension protein promoter/terminator.
Gene abbreviations: HygR, hygromycin B resistance, Sh ble, Streptoalloteichus hindustanus bleomycin/zeocin gene resistance; aphVIII, aminoglycoside 3′-phosphotransferase type VIII encoding gene from Streptomyces rimosus for paromomycin resistance.
- Abreu AP, Martins R, Nunes J. 2023. Emerging applications of Chlorella sp. and Spirulina (Arthrospira) sp. Bioeng. 10(8): 955.Article
- Adeniyi OM, Azimov U, Burluka A. 2018. Algae biofuel: Current status and future applications. Renew Sust Energ Rev. 90: 316–335. Article
- Adli M. 2018. The CRISPR tool kit for genome editing and beyond. Nat Commun. 9: 1911.ArticlePubMedPMCPDF
- Ahmad A, Jamil A, Munawar N. 2023. GMOs or non-GMOs? The CRISPR conundrum. Front Plant Sci. 14: 1232938.ArticlePubMedPMC
- Ajjawi I, Verruto J, Aqui M, Soriaga LB, Coppersmith J, et al. 2017. Lipid production in Nannochloropsis gaditana is doubled by decreasing expression of a single transcriptional regulator. Nat Biotechnol. 35(7): 647–652. ArticlePubMedPDF
- Akella S, Ma X, Bacova R, Harmer ZP, Kolackova M, et al. 2021. Co-targeting strategy for precise, scarless gene editing with CRISPR/Cas9 and donor ssODNs in Chlamydomonas. Plant Physiol. 187(4): 2637–2655. ArticlePubMedPMCPDF
- Angstenberger M, de Signori F, Vecchi V, Dall’Osto L, Bassi R. 2020. Cell synchronization enhances nuclear transformation and genome editing via Cas9 enabling homologous recombination in Chlamydomonas reinhardtii. ACS Synth Biol. 9(10): 2840–2850. ArticlePubMedPMCLink
- Aoki Y, Okamura Y, Ohta H, Kinoshita K, Obayashi T. 2016. ALCOdb: Gene coexpression database for microalgae. Plant Cell Physiol. 57(1): e3. ArticlePubMed
- Araújo R, Vázquez Calderón F, Sánchez López J, Azevedo IC, Bruhn A, et al. 2021. Current status of the algae production industry in Europe: An emerging sector of the blue bioeconomy. Front Mar Sci. 7: 626389.Article
- Baek K, Kim DH, Jeong J, Sim SJ, Melis A, et al. 2016. DNA-free two-gene knockout in Chlamydomonas reinhardtii via CRISPR-Cas9 ribonucleoproteins. Sci Rep. 6: 30620.ArticlePubMedPMCPDF
- Battarra C, Angstenberger M, Bassi R, Dall’Osto L. 2024. Efficient DNA-free co-targeting of nuclear genes in Chlamydomonas reinhardtii. Biol Direct. 19(1): 108.ArticlePubMedPMCPDF
- Bumbak F, Cook S, Zachleder V, Hauser S, Kovar K. 2011. Best practices in heterotrophic high-cell-density microalgal processes: Achievements, potential and possible limitations. Appl Microbiol Biotechnol. 91(1): 31–46. ArticlePubMedPMCPDF
- Chang KS, Kim J, Park H, Hong SJ, Lee CG, et al. 2020. Enhanced lipid productivity in agp knockout marine microalga Tetraselmis sp. using a DNA-free CRISPR-Cas9 RNP method. Bioresour Technol. 303: 122932.ArticlePubMed
- Chowdhury H, Loganathan B. 2019. Third-generation biofuels from microalgae: A review. Curr Opin Green Sustain Chem. 20: 39–44. Article
- Daboussi F, Leduc S, Maréchal A, Dubois G, Guyot V, et al. 2014. Genome engineering empowers the diatom Phaeodactylum tricornutum for biotechnology. Nat Commun. 5: 3831.ArticlePubMedPDF
- Dinesh Kumar S, Sojin K, Santhanam P, Dhanalakshmi B, Latha S, et al. 2018. Triggering of fatty acids on Tetraselmis sp. by ethyl methanesulfonate mutagenic treatment. Bioresour Technol Rep. 2: 21–28. Article
- Ferenczi A, Pyott DE, Xipnitou A, Molnar A. 2017. Efficient targeted DNA editing and replacement in Chlamydomonas reinhardtii using Cpf1 ribonucleoproteins and single-stranded DNA. Proc Natl Acad Sci USA. 114(51): 13567–13572. ArticlePubMedPMC
- Fernández FGA, Reis A, Wijffels RH, Barbosa M, Verdelho V, et al. 2021. The role of microalgae in the bioeconomy. New Biotechnol. 61: 99–107. Article
- Freudenberg RA, Wittemeier L, Einhaus A, Baier T, Kruse O. 2022. The spermidine synthase gene SPD1: A novel auxotrophic marker for Chlamydomonas reinhardtii designed by enhanced CRISPR/Cas9 gene editing. Cells. 11(5): 837.ArticlePubMedPMC
- Gaj T, Gersbach CA, Barbas CF. 2013. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 31(7): 397–405. ArticlePubMedPMC
- Görlich S, Pawolski D, Zlotnikov I, Kröger N. 2019. Control of biosilica morphology and mechanical performance by the conserved diatom gene Silicanin-1. Commun Biol. 2: 245.ArticlePubMedPMC
- Graesholt C, Brembu T, Volpe C, Bartosova Z, Serif M, et al. 2024. Zeaxanthin epoxidase 3 knockout mutants of the model diatom Phaeodactylum tricornutum enable commercial production of the bioactive carotenoid diatoxanthin. Mar Drugs. 22(4): 185.ArticlePubMedPMC
- Greiner A, Kelterborn S, Evers H, Kreimer G, Sizova I, et al. 2017. Targeting of photoreceptor genes in Chlamydomonas reinhardtii via zinc-finger nucleases and CRISPR/Cas9. Plant Cell. 29(10): 2498–2518. ArticlePubMedPMCPDF
- Guo C, Ma X, Gao F, Guo Y. 2023. Off-target effects in CRISPR/Cas9 gene editing. Front Bioeng Biotechnol. 11: 1143157.ArticlePubMedPMC
- Hao X, Chen W, Amato A, Jouhet J, Maréchal E, et al. 2022. Multiplexed CRISPR/Cas9 editing of the long-chain acyl-CoA synthetase family in the diatom Phaeodactylum tricornutum reveals that mitochondrial ptACSL3 is involved in the synthesis of storage lipids. New Phytol. 233(4): 1797–1812. ArticlePubMedLink
- Hopes A, Nekrasov V, Kamoun S, Mock T. 2016. Editing of the urease gene by CRISPR-Cas in the diatom Thalassiosira pseudonana. Plant Methods. 12: 49.ArticlePubMedPMCPDF
- Jagadevan S, Banerjee A, Banerjee C, Guria C, Tiwari R, et al. 2018. Recent developments in synthetic biology and metabolic engineering in microalgae towards biofuel production. Biotechnol Biofuels. 11: 185.ArticlePubMedPMCPDF
- Jeon MS, Han SI, Jeon M, Choi YE. 2021. Enhancement of phycoerythrin productivity in Porphyridium purpureum using the clustered regularly interspaced short palindromic repeats/CRISPR-associated protein 9 ribonucleoprotein system. Bioresour Technol. 330: 124974.ArticlePubMed
- Jiang W, Brueggeman AJ, Horken KM, Plucinak TM, Weeks DP. 2014. Successful transient expression of Cas9 and single guide RNA genes in Chlamydomonas reinhardtii. Eukaryot Cell. 13: 1465–1469. ArticlePubMedPMCLink
- Jiang WZ, Weeks DP. 2017. A gene-within-a-gene Cas9/sgRNA hybrid construct enables gene editing and gene replacement strategies in Chlamydomonas reinhardtii. Algal Res. 26: 474–480. Article
- Kang S, Jeon S, Kim S, Chang YK, Kim YC. 2020. Development of a PVEC peptide-based ribonucleoprotein (RNP) delivery system for genome editing using CRISPR/Cas9 in Chlamydomonas reinhardtii. Sci Rep. 10: 22158.ArticlePubMedPMCPDF
- Kang Z, Kim BH, Ramanan R, Choi JE, Yang JW, et al. 2015. A cost analysis of microalgal biomass and biodiesel production in open raceways treating municipal wastewater and under optimum light wavelength. J Microbiol Biotechnol. 25(1): 109–118. ArticlePubMed
- Kao PH, Ng IS. 2017. CRISPRi-mediated phosphoenolpyruvate carboxylase regulation to enhance the production of lipid in Chlamydomonas reinhardtii. Bioresour Technol. 245: 1527–1537. ArticlePubMed
- Karas BJ, Diner RE, Lefebvre SC, McQuaid J, Phillips APR, et al. 2015. Designer diatom episomes delivered by bacterial conjugation. Nat Commun. 6: 6925.ArticlePubMedPDF
- Khan MI, Shin JH, Kim JD. 2018. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Fact. 17: 36.ArticlePubMedPMCPDF
- Kilian O, Benemann CSE, Niyogi KK, Vick B. 2011. High-efficiency homologous recombination in the oil-producing alga Nannochloropsis sp. Proc Natl Acad Sci USA. 108(52): 21265–21269. ArticlePubMedPMC
- Kim J, Chang KS, Lee S, Jin E. 2021. Establishment of a genome editing tool using CRISPR-Cas9 in Chlorella vulgaris UTEX395. Int J Mol Sci. 22(2): 480.ArticlePubMedPMC
- Kim JS, Lee S, Cho S, Jung Y. 2024. Inducing heritable genomic deletions in APT gene of Chlorella sorokiniana using CRISPR/Cas9. Algal Res. 79: 103435.Article
- Kim ZH, Park YS, Ryu YJ, Lee CG. 2017. Enhancing biomass and fatty acid productivity of Tetraselmis sp. in bubble column photobioreactors by modifying light quality using light filters. Biotechnol Bioprocess Eng. 22: 397–404. ArticlePDF
- Kneip JS, Kniepkamp N, Jang J, Mortaro MG, Jin E, et al. 2024. CRISPR/Cas9-mediated knockout of the lycopene epsilon-cyclase for efficient astaxanthin production in the green microalga Chlamydomonas reinhardtii. Plants (Basel). 13(10): 1393.ArticlePubMedPMC
- Kor SD, Chowdhury N, Keot AK, Yogendra K, Chikkaputtaiah C, et al. 2023. RNA pol III promoters—key players in precisely targeted plant genome editing. Front Genet. 13: 989199.ArticlePubMedPMC
- Kouranova E, Forbes K, Zhao G, Warren J, Bartels A, et al. 2016. CRISPRs for optimal targeting: Delivery of CRISPR components as DNA, RNA, and protein into cultured cells and single-cell embryos. Hum Gene Ther. 27(6): 464–475. ArticlePubMedPMC
- Lee YY, Park R, Miller SM, Li Y. 2022. Genetic compensation of triacylglycerol biosynthesis in the green microalga Chlamydomonas reinhardtii. Plant J. 111(4): 1069–1080. ArticlePubMedPMCLink
- Leyland B, Novichkova E, Dolui AK, Jallet D, Daboussi F, et al. 2024. Acyl-CoA binding protein is required for lipid droplet degradation in the diatom Phaeodactylum tricornutum. Plant Physiol. 194(2): 958–981. ArticlePubMedPDF
- Li T, Yang Y, Qi H, Cui W, Zhang L, et al. 2023. CRISPR/Cas9 therapeutics: Progress and prospects. Signal Transduct Target Ther. 8(1): 36.ArticlePubMedPMCPDF
- Liang W, Wei L, Wang Q, You W, Poetsch A, et al. 2024. Knocking out chloroplastic aldolases/Rubisco lysine methyltransferase enhances biomass accumulation in Nannochloropsis oceanica under high-light stress. Int J Mol Sci. 25(7): 3756.ArticlePubMedPMC
- Lin JY, Lin WR, Ng IS. 2022. CRISPRa/i with adaptive single guide assisted regulation DNA (ASGARD) mediated control of Chlorella sorokiniana to enhance lipid and protein production. Biotechnol J. 17(10): e2100514.ArticlePubMedLink
- Lin WR, Ng IS. 2020. Development of CRISPR/Cas9 system in Chlorella vulgaris FSP-E to enhance lipid accumulation. Enzyme Microb Technol. 133: 109458.ArticlePubMed
- Llavero-Pasquina M, Geisler K, Holzer A, Mehrshahi P, Mendoza-Ochoa GI, et al. 2022. Thiamine metabolism genes in diatoms are not regulated by thiamine despite the presence of predicted riboswitches. New Phytol. 235(5): 1853–1867. ArticlePubMedPMCLink
- Long Y, Han X, Meng X, Xu P, Tao F. 2024. A robust yeast chassis: Comprehensive characterization of a fast-growing Saccharomyces cerevisiae. mBio. 15(2): e0319623. ArticlePubMedLink
- Lu Y, Zhang X, Gu X, Lin H, Melis A. 2021. Engineering microalgae: Transition from empirical design to programmable cells. Crit Rev Biotechnol. 41(8): 1233–1256. ArticlePubMed
- Ma Y, Wang Z, Zhu M, Yu C, Cao Y, et al. 2013. Increased lipid productivity and TAG content in Nannochloropsis by heavy-ion irradiation mutagenesis. Bioresour Technol. 136: 360–367. ArticlePubMed
- Macdonald Miller S, Abbriano RM, Herdean A, Banati R, Ralph PJ, et al. 2023. Random mutagenesis of Phaeodactylum tricornutum using ultraviolet, chemical, and X-ray irradiation demonstrates the need for temporal analysis of phenotype stability. Sci Rep. 13(1): 22385.ArticlePubMedPMC
- Mallick N, Bagchi SK, Koley S, Singh AK. 2016. Progress and challenges in microalgal biodiesel production. Front Microbiol. 7: 1019.ArticlePubMedPMC
- Manandhar-Shrestha K, Hildebrand M. 2013. Development of flow cytometric procedures for the efficient isolation of improved lipid accumulation mutants in a Chlorella sp. microalga. J Appl Phycol. 25(6): 1643–1651. ArticlePubMedPMCPDF
- Mohanraju P, Van Der Oost J, Jinek M, Swarts DC. 2018. Heterologous expression and purification of CRISPR-Cas12a/Cpf1. Bio Protoc. 8(9): e2842. ArticlePubMedPMC
- Naduthodi MIS, Mohanraju P, Südfeld C, D’Adamo S, Barbosa MJ, et al. 2019. CRISPR-Cas ribonucleoprotein mediated homology-directed repair for efficient targeted genome editing in microalgae Nannochloropsis oceanica IMET1. Biotechnol Biofuels. 12: 66.ArticlePubMedPMCPDF
- Naduthodi MIS, Sudfeld C, Avitzigiannis EK, Trevisan N, van Lith E, et al. 2021. Comprehensive genome engineering toolbox for microalgae Nannochloropsis oceanica based on CRISPR-Cas systems. ACS Synth Biol. 10(12): 3369–3378. ArticlePubMedPMCLink
- Nawaly H, Tsuji Y, Matsuda Y. 2020. Rapid and precise genome editing in a marine diatom, Thalassiosira pseudonana by Cas9 nickase (D10A). Algal Res. 47: 101855.Article
- Nguyen THT, Park S, Jeong J, Shin YS, Sim SJ, et al. 2020. Enhancing lipid productivity by modulating lipid catabolism using the CRISPR-Cas9 system in Chlamydomonas. J Appl Phycol. 32: 2829–2840. ArticlePDF
- Nomura T, Inoue K, Uehara-Yamaguchi Y, Yamada K, Iwata O, et al. 2019. Highly efficient transgene-free targeted mutagenesis and single-stranded oligodeoxynucleotide-mediated precise knock-in in the industrial microalga Euglena gracilis using Cas9 ribonucleoproteins. Plant Biotechnol J. 17(11): 2032–2034. ArticlePubMedPMCLink
- Nomura T, Kim JS, Ishikawa M, Suzuki K, Mochida K. 2024. High-efficiency genome editing by Cas12a ribonucleoprotein complex in Euglena gracilis. Microb Biotechnol. 17(2): e14393. ArticlePubMedPMCLink
- Nymark M, Finazzi G, Volpe C, Serif M, Fonseca DM, et al. 2023. Loss of CpFTSY reduces photosynthetic performance and affects insertion of PsaC of PSI in diatoms. Plant Cell Physiol. 64(6): 583–603. ArticlePubMedPMCPDF
- Nymark M, Sharma AK, Hafskjold MCG, Sparstad T, Bones AM, et al. 2017. CRISPR/Cas9 gene editing in the marine diatom Phaeodactylum tricornutum. Bio Protoc. 7(15): e2442. ArticlePubMedPMC
- Nymark M, Sharma AK, Sparstad T, Bones AM, Winge P. 2016. A CRISPR/Cas9 system adapted for gene editing in marine algae. Sci Rep. 6: 24951.ArticlePubMedPMCPDF
- Occhipinti PS, Russo N, Foti P, Zingale IM, Pino A, et al. 2024. Current challenges of microalgae applications: Exploiting the potential of non-conventional microalgae species. J Sci Food Agric. 104(7): 3823–3833. ArticlePubMed
- Onyeaka H, Miri T, Obileke K, Hart A, Anumudu C, et al. 2021. Minimizing carbon footprint via microalgae as a biological capture. Carbon Capture Sci Technol. 1: 100007.Article
- Ortiz-Matamoros MF, Villanueva MA, Islas-Flores T. 2017. Genetic transformation of cell-walled plant and algae cells: Delivering DNA through the cell wall. Brief Funct Genomics. 17(1): 26–33. Article
- Patel VK, Das A, Kumari R, Kajla S. 2023. Recent progress and challenges in CRISPR-Cas9 engineered algae and cyanobacteria. Algal Res. 71: 103068.Article
- Picariello T, Hou Y, Kubo T, McNeill NA, Yanagisawa HA, et al. 2020. TIM, a targeted insertional mutagenesis method utilizing CRISPR/Cas9 in Chlamydomonas reinhardtii. PLoS One. 15(5): e0232594. ArticlePubMedPMC
- Pittman JK, Dean AP, Osundeko O. 2011. The potential of sustainable algal biofuel production using wastewater resources. Bioresour Technol. 102(1): 17–25. ArticlePubMed
- Plecenikova A, Mages W, Andrésson ÓS, Hrossova D, Valuchova S, et al. 2013. Studies on recombination processes in two Chlamydomonas reinhardtii endogenous genes, NIT1 and ARG7. Protist. 164(4): 570–582. ArticlePubMed
- Poliner E, Takeuchi T, Du ZY, Benning C, Farré EM. 2018. Nontransgenic marker-free gene disruption by an episomal CRISPR system in the oleaginous microalga, Nannochloropsis oceanica CCMP1779. ACS Synth Biol. 7(4): 962–968. ArticlePubMed
- Qin S, Wang K, Gao F, Ge B, Cui H, et al. 2023. Biotechnologies for bulk production of microalgal biomass: From mass cultivation to dried biomass acquisition. Biotechnol Biofuels Bioprod. 16(1): 131.ArticlePubMedPMCPDF
- Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, et al. 2013. Genome engineering using the CRISPR-Cas9 system. Nat Protoc. 8: 2281–2308. ArticlePubMedPMCPDF
- Rastogi A, Vieira FRJ, Deton-Cabanillas AF, Veluchamy A, Cantrel C, et al. 2020. A genomics approach reveals the global genetic polymorphism, structure, and functional diversity of ten accessions of the marine model diatom Phaeodactylum tricornutum. ISME J. 14(2): 347–363. ArticlePubMedPDF
- Rozenberg JM, Sorokin BA, Mukhambetova AN, Emelianova AA, Kuzmin VV, et al. 2024. Recent advances and fundamentals of microalgae cultivation technology. Biotechnol J. 19(3): e2300725. ArticlePubMed
- Russo MT, Aiese Cigliano R, Sanseverino W, Ferrante MI. 2018. Assessment of genomic changes in a CRISPR/Cas9 Phaeodactylum tricornutum mutant through whole genome resequencing. PeerJ. 6: e5507. ArticlePubMedPMCPDF
- Ryu AJ, Jeong BR, Kang NK, Jeon S, Sohn MG, et al. 2021. Safe-harboring based novel genetic toolkit for Nannochloropsis salina CCMP1776: Efficient overexpression of transgene via CRISPR/Cas9-mediated knock-in at the transcriptional hotspot. Bioresour Technol. 340: 125676.ArticlePubMed
- Sandor R, Wagh SG, Kelterborn S, Großkinsky DK, Novak O, et al. 2024. Cytokinin-deficient Chlamydomonas reinhardtii CRISPR-Cas9 mutants show reduced ability to prime resistance of tobacco against bacterial infection. Physiol Plant. 176(3): e14311. ArticlePubMed
- Schmid GH, Schwarze P. 1969. Blue light enhanced respiration in a colorless Chlorella mutant. Biol Chem. 350(12): 1513–1520. Article
- Serif M, Dubois G, Finoux AL, Teste MA, Jallet D, et al. 2018. One-step generation of multiple gene knock-outs in the diatom Phaeodactylum tricornutum by DNA-free genome editing. Nat Commun. 9(1): 3924.ArticlePubMedPMCPDF
- Shin YS, Jeong J, Nguyen THT, Kim JYH, Jin E, et al. 2019. Targeted knockout of phospholipase A2 to increase lipid productivity in Chlamydomonas reinhardtii for biodiesel production. Bioresour Technol. 271: 368–374. ArticlePubMed
- Shin SE, Lim JM, Koh HG, Kim EK, Kang NK, et al. 2016. CRISPR/Cas9-induced knockout and knock-in mutations in Chlamydomonas reinhardtii. Sci Rep. 6: 27810.ArticlePubMedPMCPDF
- Silva G, Poirot L, Galetto R, Smith J, Montoya G, et al. 2011. Meganucleases and other tools for targeted genome engineering: Perspectives and challenges for gene therapy. Curr Gene Ther. 11(1): 11–27. ArticlePubMedPMC
- Sizova I, Greiner A, Awasthi M, Kateriya S, Hegemann P. 2013. Nuclear gene targeting in Chlamydomonas using engineered zinc-finger nucleases. Plant J. 73(5): 873–882. ArticlePubMed
- Song I, Kim J, Baek K, Choi Y, Shin B, et al. 2020. The generation of metabolic changes for the production of high-purity zeaxanthin mediated by CRISPR-Cas9 in Chlamydomonas reinhardtii. Microb Cell Fact. 19: 220.ArticlePubMedPMCPDF
- Song I, Kim S, Kim J, Oh H, Jang J, et al. 2022. Macular pigment-enriched oil production from genome-edited microalgae. Microb Cell Fact. 21: 27.ArticlePubMedPMCPDF
- Tamaki S, Ozasa K, Nomura T, Ishikawa M, Yamada K, et al. 2023. Zeaxanthin is required for eyespot formation and phototaxis in Euglena gracilis. Plant Physiol. 191(4): 2414–2426. ArticlePubMedPMCPDF
- Teng CS, Ng IS. 2023. Optimization of 4-aminobutyric acid feeding strategy and clustered regularly interspaced short palindromic repeats activation for enhanced value-added chemicals in halophilic Chlorella sorokiniana. Bioresour Technol. 387: 129599.ArticlePubMed
- Trovão M, Schüler LM, Machado A, Bombo G, Navalho S, et al. 2022. Random mutagenesis as a promising tool for microalgal strain improvement towards industrial production. Mar Drugs. 20(7): 440.ArticlePubMedPMC
- Tsai SQ, Joung JK. 2016. Defining and improving the genome-wide specificities of CRISPR-Cas9 nucleases. Nat Rev Genet. 17(5): 300–312. ArticlePubMedPMCPDF
- Venkatesan J, Manivasagan P, Kim SK. 2015. Marine microalgae biotechnology: Present trends and future advances. In Kim SK (ed.), Handbook of marine microalgae, pp. 1-9. Academic Press. Article
- Verruto J, Francis K, Wang Y, Low MC, Greiner J, et al. 2018. Unrestrained markerless trait stacking in Nannochloropsis gaditana through combined genome editing and marker recycling technologies. Proc Natl Acad Sci USA. 115(30): E7015–E7022. ArticlePubMedPMC
- Waltz E. 2016. Gene-edited CRISPR mushroom escapes US regulation. Nature. 532: 293.ArticlePubMedPDF
- Wang Q, Gong Y, He Y, Xin Y, Lv N, et al. 2021. Genome engineering of Nannochloropsis with hundred-kilobase fragment deletions by Cas9 cleavages. Plant J. 106(4): 1148–1162. ArticlePubMedLink
- Wang Q, Lu Y, Xin Y, Wei L, Huang S, et al. 2016. Genome editing of model oleaginous microalgae Nannochloropsis spp. by CRISPR/Cas9. Plant J. 88(6): 1071–1081. ArticlePubMed
- Wang H, Slattery SS, Karas BJ, Edgell DR. 2018. Delivery of the Cas9 or tevCas9 system into Phaeodactylum tricornutum via conjugation of plasmids from a bacterial donor. Bio Protoc. 8(16): e2974. ArticlePubMedPMC
- Wei L, Jiang Z, Liu B. 2022. A CRISPR/dCas9-based transcription activated system developed in marine microalga Nannochloropsis oceanica. Aquaculture. 546: 737064.Article
- Xie X, Yang J, Du H, Chen J, Sanganyado E, et al. 2023. Golgi fucosyltransferase 1 reveals its important role in α-1,4-fucose modification of N-glycan in CRISPR/Cas9 diatom Phaeodactylum tricornutum. Microb Cell Fact. 22(1): 6.ArticlePubMedPMCPDF
- Yang W, Zhou L, Wang J, Wang L, Gao S, et al. 2022. Knockout of a diatom cryptochrome by CRISPR/Cas9 causes an increase in light-harvesting protein levels and accumulation of fucoxanthin. Algal Res. 66: 102822.Article
- Ye C, Mu D, Horowitz N, Xue Z, Chen J, et al. 2018. Life cycle assessment of industrial scale production of Spirulina tablets. Algal Res. 34: 154–163. Article
- Yoneda K, Oishi R, Yoshida M, Matsuda Y, Suzuki I. 2023. Stramenopile-type lipid droplet protein functions as a lipid droplet scaffold protein in the marine diatom Phaeodactylum tricornutum. Plant Cell Physiol. 64(7): 803–813. ArticlePubMedPDF
- Yu J, Baek K, Jin E, Bae S. 2017. DNA-free genome editing of Chlamydomonas reinhardtii using CRISPR and subsequent mutant analysis. Bio Protoc. 7(11): e2352. ArticlePubMedPMC
- Zadabbas Shahabadi H, Akbarzadeh A, Ofoghi H, Kadkhodaei S. 2023. Site-specific gene knock-in and bacterial phytase gene expression in Chlamydomonas reinhardtii via Cas9 RNP-mediated HDR. Front Plant Sci. 14: 1150436.ArticlePubMedPMC
- Zhang B, Wu J, Meng F. 2021. Adaptive laboratory evolution of microalgae: A review of the regulation of growth, stress resistance, metabolic processes, and biodegradation of pollutants. Front Microbiol. 12: 737248.ArticlePubMedPMC
- Zhang Z, Xu M, Fan Y, Zhang L, Wang H. 2024. Using microalgae to reduce the use of conventional fertilizers in hydroponics and soil-based cultivation. Sci Total Environ. 912: 169424.ArticlePubMed
References
Supplementary Information
References
Citations

- Active and targeted micro/nanoplastics remediation via engineered microalgae co-displaying polymer-binding peptides and plastic-degrading enzymes: A critical review and perspectives
Ling Wang, Mingjing Zhang, Jialin Wang, Chen Hu, Zhanyou Chi, Lei Li, Wenjun Luo, Chengze Li, Chenba Zhu
Algal Research.2026; 93: 104455. CrossRef - Advancing microbial engineering through synthetic biology
Ki Jun Jeong
Journal of Microbiology.2025; 63(3): e2503100. CrossRef - Progress and prospects in metabolic engineering approaches for isoprenoid biosynthesis in microalgae
Sonia Mohamadnia, Borja Valverde-Pérez, Omid Tavakoli, Irini Angelidaki
Biotechnology for Biofuels and Bioproducts.2025;[Epub] CrossRef - Beyond Biomass: Reimagining Microalgae as Living Environmental Nano-Factories
Thinesh Selvaratnam, Shaseevarajan Sivanantharajah, Kirusha Sriram
Environments.2025; 12(7): 221. CrossRef - Harnessing MicroRNAs and CRISPR to enhance biofuel production in microalgae
Dariga K. Kirbayeva, Altynay Y. Shayakhmetova, Bekzhan D. Kossalbayev, Assemgul K. Sadvakasova, Meruyert O. Bauenova
International Journal of Hydrogen Energy.2025; 157: 150399. CrossRef - Beyond Cutting: CRISPR-Driven Synthetic Biology Toolkit for Next-Generation Microalgal Metabolic Engineering
Limin Yang, Qian Lu
International Journal of Molecular Sciences.2025; 26(15): 7470. CrossRef - Mechanistic Role of Heavy Metals in Driving Antimicrobial Resistance: From Rhizosphere to Phyllosphere
Rahul Kumar, Tanja P. Vasić, Sanja P. Živković, Periyasamy Panneerselvam, Gustavo Santoyo, Sergio de los Santos Villalobos, Adeyemi Nurudeen Olatunbosun, Aditi Pandit, Leonard Koolman, Debasis Mitra, Pankaj Gautam
Applied Microbiology.2025; 5(3): 79. CrossRef - Strain Improvement Through Genetic Engineering and Synthetic Biology for the Creation of Microalgae with Enhanced Lipid Accumulation, Stress Tolerance, and Production of High-value
Alebachew Molla, Gedif Meseret
Science Frontiers.2025; 6(3): 80. CrossRef - The Role of Molecular Tools in Microalgal Strain Improvement: Current Status and Future Perspectives
Alebachew Molla, Gedif Meseret
Advances in Bioscience and Bioengineering.2025; 13(3): 51. CrossRef - CRISPR-Cas9 genome editing in microalgae for improved high-value products (HVP) production
Fazleen Haslinda Mohd Hatta, Nurin Nisa’ Ahmad Zamri, Norazlina Ahmad
Asia Pacific Journal of Molecular Biology and Biotechnology.2025; : 245. CrossRef - Advances in Algae-Based Bioplastics: From Strain Engineering and Fermentation to Commercialization and Sustainability
Nilay Kumar Sarker, Prasad Kaparaju
Fermentation.2025; 11(10): 574. CrossRef - Exploring the nutritional and bioactive potential of microalgal sulfated polysaccharides for functional food applications
Fajar Sofyantoro, Eka Sunarwidhi Prasedya, Fahrul Nurkolis, Andri Frediansyah
Food Science and Biotechnology.2025;[Epub] CrossRef - Harnessing microalgae for bioproducts: innovations in synthetic biology
Zheng Li, Yuhui Cheng, Chengcheng Li, Qianyi Wu, Yi Xin
World Journal of Microbiology and Biotechnology.2025;[Epub] CrossRef
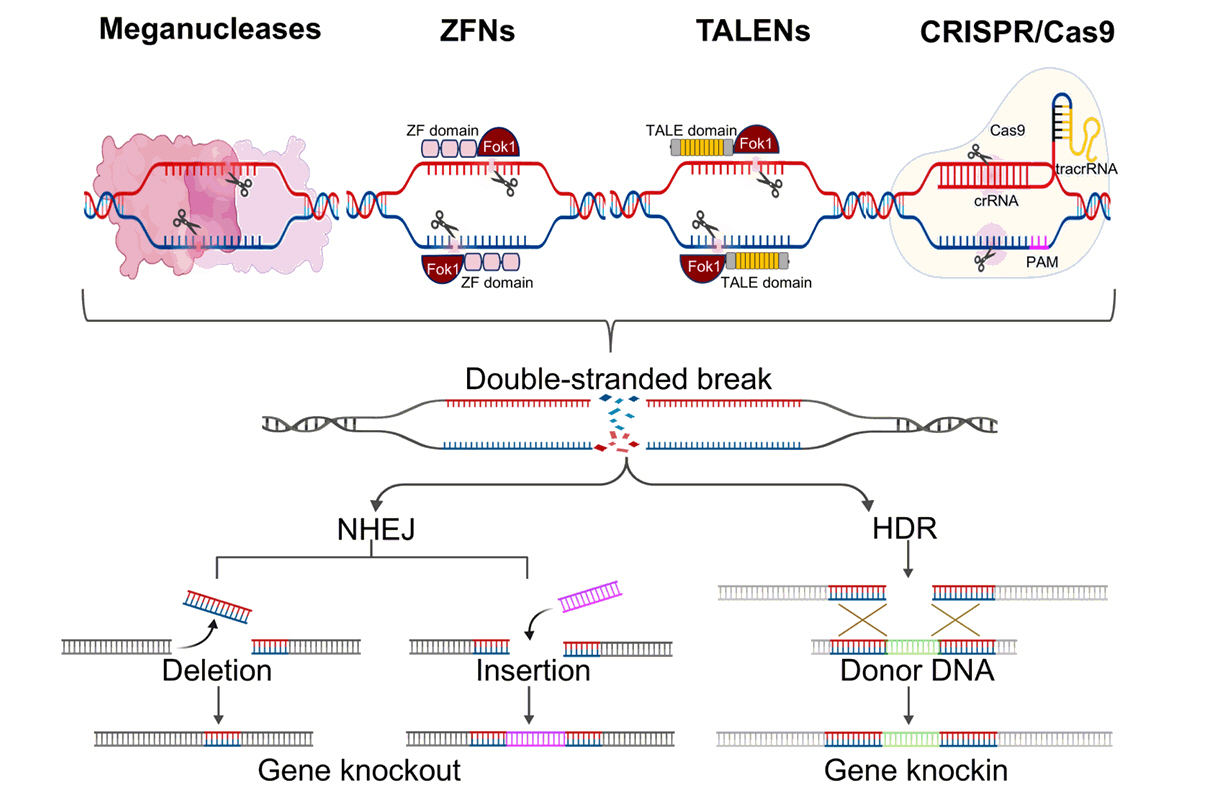
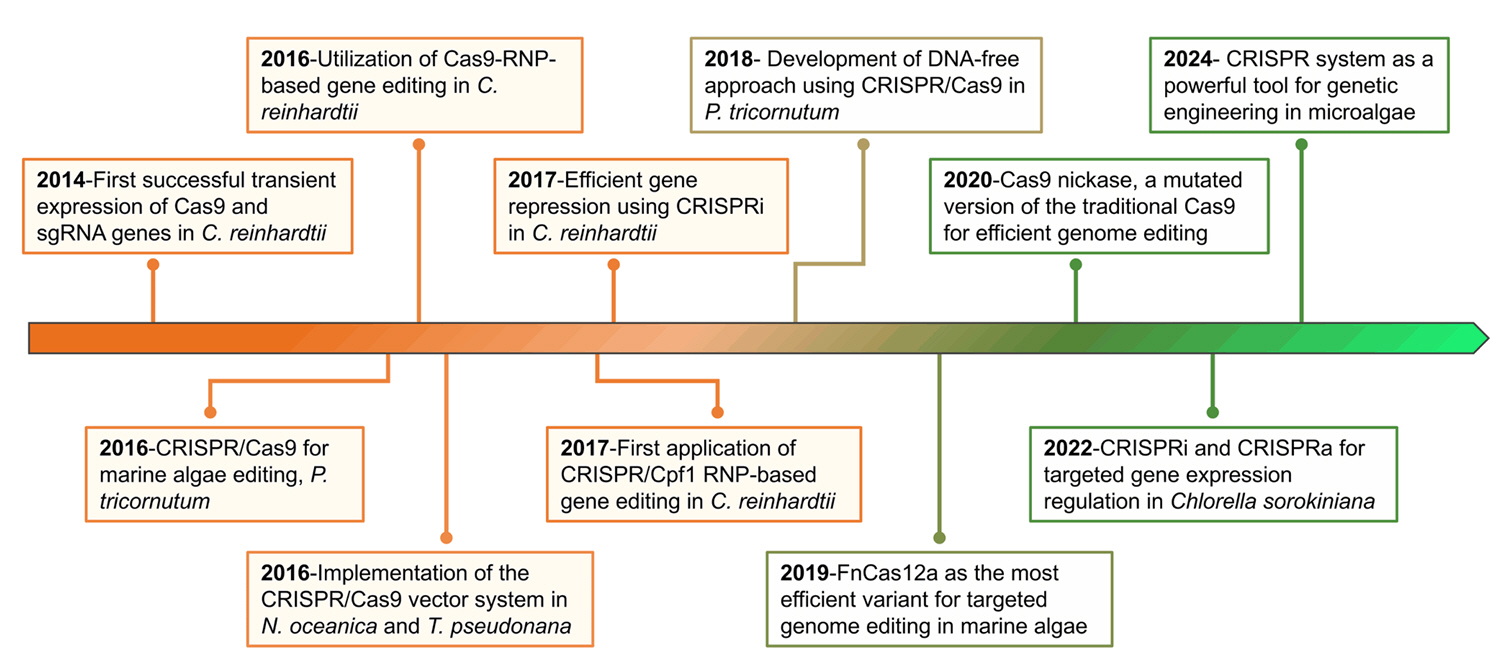
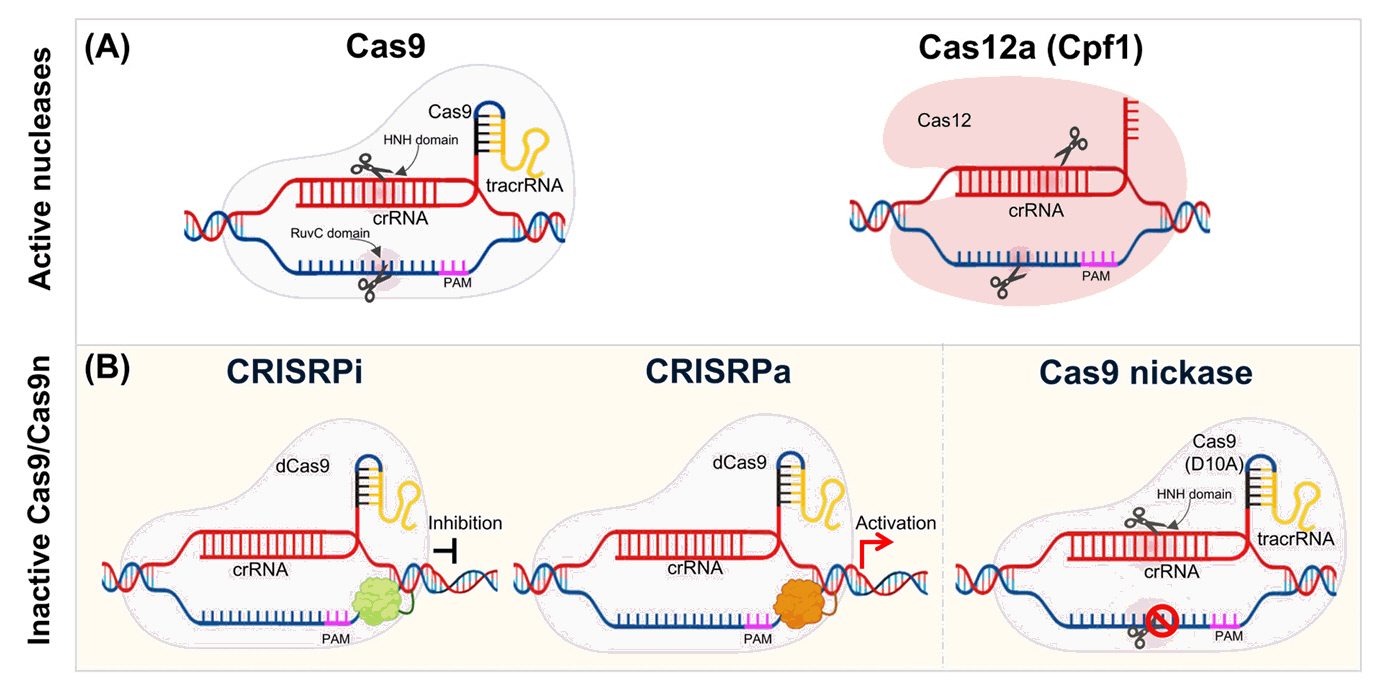
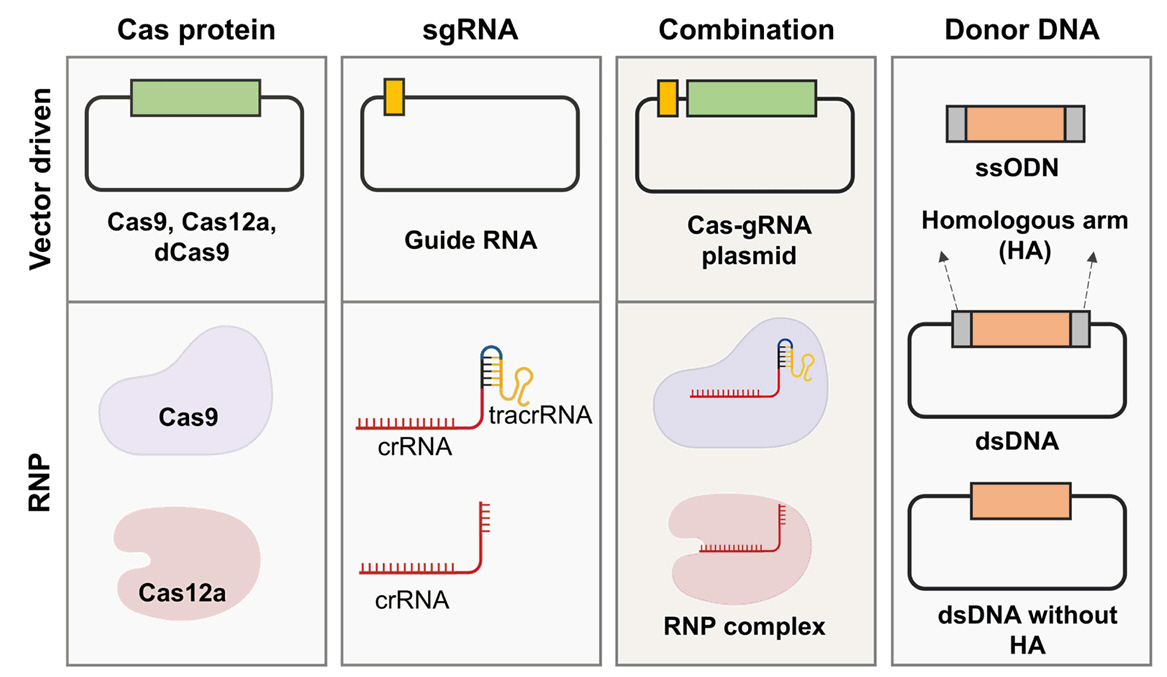
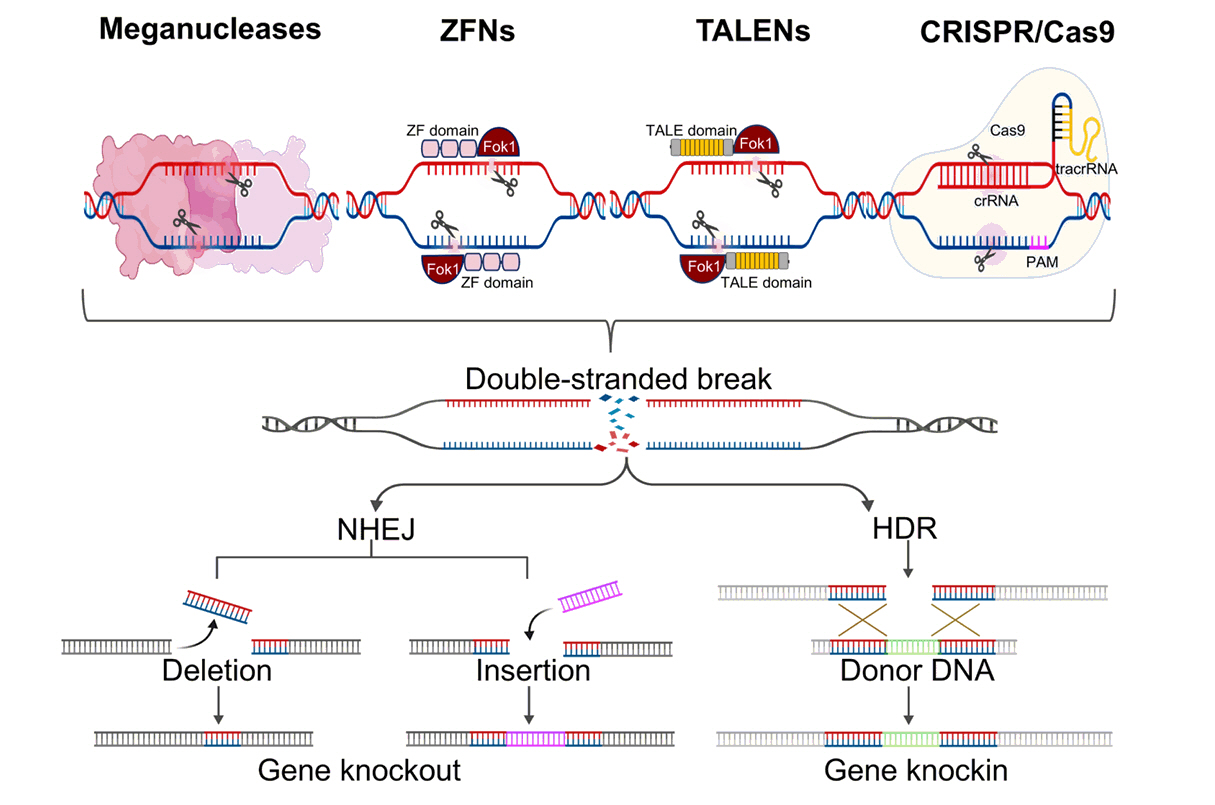
Fig. 1.
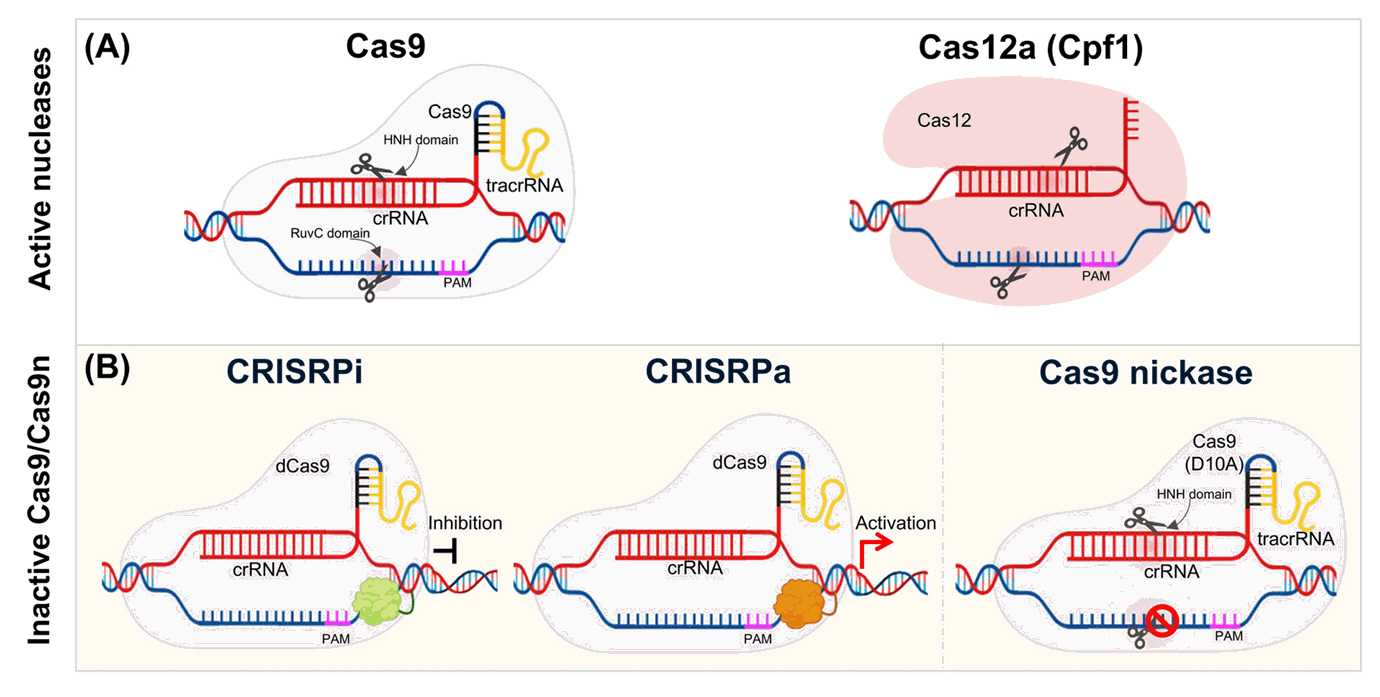
Fig. 2.
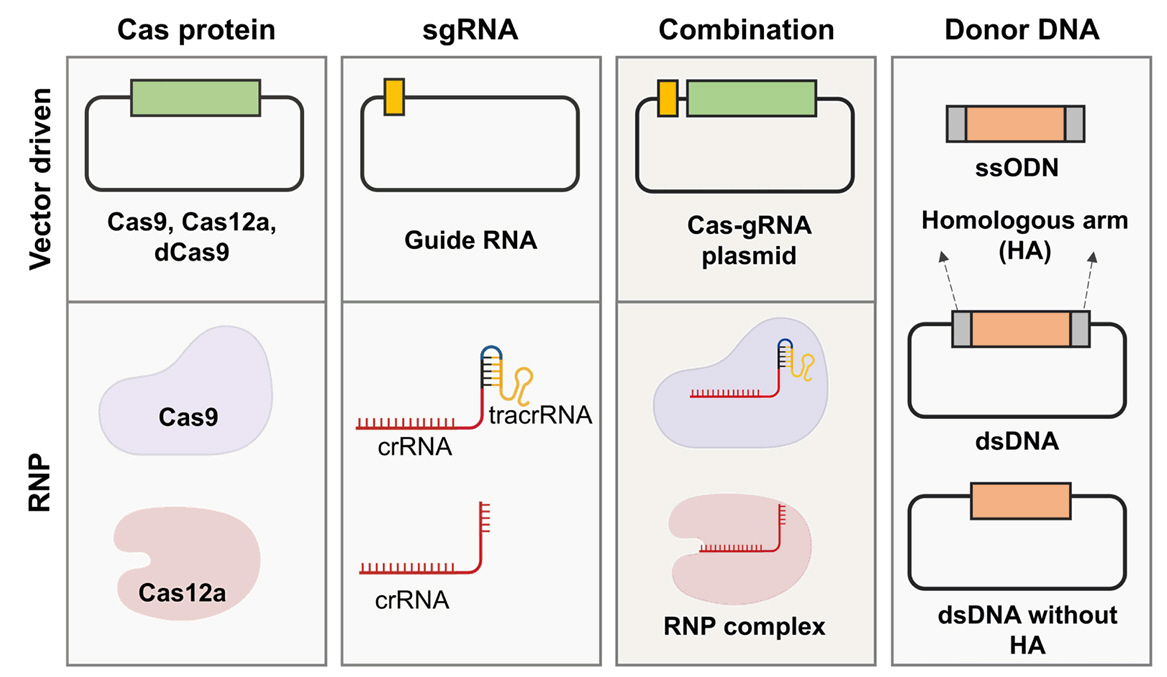
Fig. 3.
Fig. 4.
| Algal strain | CRISPR/Cas system | Strategy |
Targeted genes | Selection Marker | Products | Mutagenesis efficiency (%) |
Targeted mutagenesis frequency |
References | |
|---|---|---|---|---|---|---|---|---|---|
| Approaches | Delivery method | ||||||||
| Chlamydomonas reinhardtii | Cas9 | Vector driven | Electroporation | FKB12 | Rapamycin | First successful transient expression of Cas9 and sgRNA genes in C. reinhardtii | 0.0014 | 2 × 10-8 | |
| Cas9 | RNP | Electroporation | MAA7, CpSRP43, ChlM | Auxotrophic selection using 5-FI | Visible auxotrophic colonies with mutations targeted at the Cas9 cut sites | 40% (MAA7), 1.4% (CpSRP43), and 0.17% (ChlM) | 8.9 × 10-8 (MAA7), 3.3 × 10-8 (CpSRP43), and 5 × 10-8 (ChlM) | ||
| Cas9 | RNP | Electroporation | CpFTSY, ZEP | Based on the coloration of the cells | High zeaxanthin-producing mutants with improved photosynthetic productivity | 0.46 to 0.56% | Not mention | ||
| CRISPRi | Vector driven | Glass beads | PEPC1 | Hygromycin and paromomycin | A 74.4% increase in lipid content and a 94.2% enhancement in lipid productivity | 0.94 | Not mention | ||
| Cas9 | Vector driven/RNP | Electroporation | COP1/2, COP3, COP4, COP5, PHOT, UVR8, VGCC, MAT3, aCRY, PSY1 | Paromomycin | Protocols for the rapid isolation of non-selectable gene mutants | 5 to 15% | 2.5 × 10˗5 to 2.5 × 10-4 | ||
| Cas9 | Vector driven/RNP | Electroporation | FKB12, ALS, ARG | Rapamycin and zeocin | Gene-within-a-gene hybrid construct, composed of a Cas9 gene containing an artificial intron and an inserted sgRNA gene | Not mention | 3 × 10-8 | ||
| Cpf1 (Cas12a) | RNP | Electroporation | FKB12, CpFTSY, CpSRP43, PHT7 | Rapamycin and based on coloration and chlorophyll fluorescence | CRISPR/Cpf1-mediated DNA editing efficiencies increased 500-fold with the use of single-stranded oligodeoxynucleotides (ssODNs) | ∼10% | Not mention | ||
| Cas9 | RNP | Electroporation | PLA2 | Hygromycin | Lipid productivity in phospholipase A2 knockout mutants increased by 64.25%, reaching 80.92 g/L/d | Not mention | Not mention | ||
| Cas9 | RNP | Electroporation | ELT1 | Hygromycin | Total lipid accumulated up to 28% of dry biomass, with a 27.2% increase in C18:1 ratio | 0.1368 | Not mention | ||
| Cas9 | RNP | Electroporation | LCYE, ZEP | Hygromycin | Mutant with 60% higher zeaxanthin yield (5.24 mg/L) and content (7.28 mg/g) | Not mention | Not mention | ||
| Cas9 | RNP | Electroporation and glass beads | IFT81, FAP70, MOT17, CDPK13, CEP131 | Paromomycin | Development of CRISPR-based targeted insertional mutagenesis method (TIM) for C. reinhardtii | 40% to 95% | Not mention | ||
| Cas9 | RNP | Cell-penetrating peptide pVEC | Maa7, FKB12 | Auxotrophic selection using 5-FI (MAA7) and Rapamycin (FKB12) | Delivery of Cas9/sgRNA RNP into C. reinhardtii using cell-penetrating peptide pVEC | 8.41% to 46.56% | Not mention | ||
| Cas9 | RNP | Electroporation | PPX1, FTSY, WDTC1 | Paromomycin or oxyfluorfen | Generation of individual strains with precise mutations in multiple target genes | 1.3% (PPX1 and FTSY) and 0.8% (WDTC1) | Not mention | ||
| Cas9 | RNP | Electroporation | SPD1 | Paromomycin or hygromycin | Targeted knockout of CrSPD1 induces spermidine auxotroph, which could be used as a selectable marker in biotechnology | 10% to 66% | Not mention | ||
| Cas9 | RNP | Glass bead | NR | Hygromycin | Generation of mutants with a bacterial phytase gene cassette knocking into the NR gene | 0.1481 | Not mention | ||
| Cas9 | RNP | Electroporation | PSR1, CpFTSY | 30% starch | Generation of mutants with impaired extracellular phosphatase synthesis in response to Pi deprivation | 0.22 | Not mention | ||
| Cas9 | RNP | Electroporation | LCYE | Hygromycin | A 2.3-fold increase in astaxanthin accumulation in the ΔLCYE mutant | 0.0417 | Not mention | ||
| Nannochloropsis spp. | Cas9 | Vector driven | Electroporation | NR | Grow normally under NH4Cl but fail to grow under NaNO3 | Knockout of the NR gene | 0.1% to 1% | Not mention | |
| Cas9 | In vitro–synthesized gRNAs/Cas9-expressing line | Electroporation | Insertional mutagenesis of 18 gene of 20 transcription factors | Blasticidin or Hygromycin | Improvement in the total carbon-to-lipid ratio from 20% (wild type) to 40–55% (mutants) | 6.25 to 78% | Not mention | ||
| Cas9 | In vitro–synthesized gRNAs/reporter-free Cas9-expressing line | Electroporation | Aco1 | Blasticidin and Hygromycin | Mutants with doubled lipid productivity and ∼50% reduced photosynthetic antenna size | ∼50% | Not mention | ||
| Cas9 and Cas12a | RNP | Electroporation | NR | Zeocin | FnCas12a generated HDR-based mutants with up to 93% efficiency | 34–71% (for Cas9) and 3-93% (for Cas12a) | Not mention | ||
| Cas9 | In vitro–synthesized gRNAs/reporter-free Cas9- expressing line | Electroporation | Knock-in of FAD12 gene at the T1 hotspot | Zeocin | Improved production of polyunsaturated fatty acids (PUFAs) | 0.714 | Not mention | ||
| Cas12a | RNP | Electroporation | NR | Grow normally under NH4Cl but fail to grow under NaNO3 | Generation of markerless genome editing tool to knock out target genes | 0.1052 | Not mention | ||
| Cas9 | Vector driven | Electroporation | LER1 and LER2 | Hygromycin | Double deletion of both LER1 and LER2 (from chromosome 9), total ~214 kb | 0.285 | Not mention | ||
| dCas9 | Vector driven | Electroporation | g1248 | Zeocin | Growth and photosynthetic parameters (Fv/Fm) of the mutants increased by 23% and 12%, respectively, compared to the wild type under ambient CO2 levels | Not mention | Not mention | ||
| Cas9 | Vector driven | Bombardment | LSMT | Hygromycin | Mutation induced 18–20% reduction in fructose-1,6-bisphosphate aldolases, along with 9.7–13.8% increase in dry weight and enhanced growth | 0.1 | 2 × 10-8 | ||
| Phaeodactylum tricornutum | Cas9 | Vector driven | Bombardment | CpSRP54 | Zeocin | Generation of stable targeted gene mutations in marine algae | 0.31 | Not mention | |
| Cas9 | Vector-driven and RNP | Bombardment | PtUMPS, PtAPT, PtAureo1a | Nourseothricin, 2-FA, adenine, and uracil | A single-step generation of triple knockout strains | 65% to 100% | Not mention | ||
| Cas9 | Vector driven | Bacterial conjugation | Phatr3_J46193 | Phleomycin | Bacterial conjugation-mediated Cas9 delivery to minimize genome exposure to nuclease activity | Not mention | 2 × 10-5 | ||
| Cas9 | Vector driven | Electroporation | LACS | Zeocin | Reduced growth rate and altered molecular profiles of PC and TAGs | Not mention | Not mention | ||
| Cas9 | Vector driven | Conjugation of plasmids | CryP | Zeocin | Increased light-harvesting protein levels in CryP knockout mutants | Not mention | Not mention | ||
| Cas9 | Vector driven | Bombardment | PtTHIC and PtSSSP | Zeocin | Targeted mutation of the TPP aptamer in the THIC gene encoding HMP-P synthase does not affect thiamine biosynthesis in P. tricornutum | Not mention | Not mention | ||
| Cas9 | Vector driven | Bombardment | CpFTSY | Zeocin | Generation and characterization of CpFTSY mutants | Not mention | Not mention | ||
| Cas9 | Vector driven | Bombardment | FucT | Zeocin | Knockout of PtFucT1 affected PtGnTI activity in the complex, converting the N-glycan to a mannose-type N-glycan | 0.333 | Not mention | ||
| Cas9 | Vector driven | Electroporation | StLDP | Zeocin | StLDP functions as an LD scaffold protein in P. tricornutum, regulating LD numbers in the stldp mutant and complemented strains | 0.8125 | Not mention | ||
| Cas9 | Vector driven | Bacterial conjugation | ZEP 2, ZEP3 | No selective pressure | Generation of zep mutants as a platform for diatoxanthin production | Not mention | Not mention | ||
| Chlorella sp. | Cas9 | Vector driven | Electroporation | fad3 | Hygromycin | Mutants have 46% higher lipid accumulation | Not mention | Not mention | |
| Cas9 | Vector driven and RNP | Bombardment | NR, APT | NaNO2 and KClO3 | Generation of auxotrophic strains | Not mention | Not mention | ||
| CRISPRi and CRISPRa | Vector driven | Electroporation | Randomly mediate gene regulation | Hygromycin | Mutants with protein content of 60% to 65% (w/w) of dry cell weight | 0.5 | Not mention | ||
| Cas9 | Vector driven | Electroporation | GS | Hygromycin | Generation of mutants with enhanced biomass, protein, and lutein content | Not mention | Not mention | ||
| Cas9 | Vector driven | Electroporation | APT | Hygromycin | Combination of Alcalase treatment with PEG transformation for efficient gene editing in Chlorella | Not mention | 1 × 10-7 to 2 × 10-7 | ||
| Porphyridium purpureum | Cas9 | RNP | Bombardment | CHS1 | No selective pressure | Generation of chlorophyll synthase loss-of-function mutants with increased phycoerythrin levels | Not mention | Not mention | |
| Tetraselmis sp. | Cas9 | RNP | Bombardment | AGP | No selective pressure | Mutants had 2.7- and 3.1-fold increased lipid content (21.1% and 24.1% of DCW, respectively) | Not mention | ||
| Euglena gracilis | Cas9 | RNP | Electroporation | EgGSL2 | No selective pressure | Transgene-free targeted mutagenesis and ssODN-mediated gene knockin | 77.7 to 90.1% | Not mention | |
| LbCas12a | RNP | Electroporation | EgGSL2, EgcrtB | No selective pressure | High-efficiency genome editing system using direct delivery of LbCas12a RNP complexes | 77.2–94.5% | Not mention | ||
| Cas9 | RNP | Electroporation | Knockout of 16 carotenoid biosynthetic genes present in E. gracilis | No selective pressure | Mutants with different carotenoid compositions | Not mention | Not mention | ||
| Thalassiosira pseudonana | Cas9 | Vector driven | Bombardment | Urease | Nourseothricin | Significant reduction in growth rate and cell size compared to nitrate growth | 0.121 | 8 × 10-7 | |
| Cas9 | Vector driven | Bombardment | Sin1 | Nourseothricin | Mutants exhibit reduced biosilica content and morphological aberrations, affecting cell wall strength and stiffness | Not mention | Not mention | ||
| Cas9 nickase | Vector driven | Bombardment | TpθCA3 | Nourseothricin | Development of an efficient Cas9 nickase (D10A) system for highly specific indel introduction into target DNA | 0.39 | 3.4 × 10-7 | ||
| Components | Source/available toolkits | Company |
|---|---|---|
| Plasmids | Addgene (USA) | |
| SnapGene Plasmid Database (USA) | ||
| GenScript (USA) | ||
| IDT (USA) | ||
| Cas9 | ToolGen, Inc. (Korea) | |
| Macrogen, Inc. (Korea) | ||
| IDT (USA) | ||
| NEB (USA) | ||
| TaKaRa (Japan) | ||
| gRNA | MEGAshortscriptTM T7 Kit | Ambion (USA) |
| HiScribe T7 RNA Kit | NEB (USA) | |
| CUGA7 gRNA Synthesis Kit | Nippon Gene (Japan) | |
| EnGen® sgRNA Synthesis Kit | NEB (USA) | |
| Guide-itTM sgRNA In Vitro Transcription | TaKaRa (Japan) |
| Microalgal strains | Variants of Cas protein | Origin | Codon optimization | PAM | References |
|---|---|---|---|---|---|
| C. reinhardtii | SpCas9 | Streptococcus pyogenes | Yes | NGG | |
| C. reinhardtii | SaCas9 and SpCas9 | Staphylococcus aureus and S. pyogenes | Yes | NGG | |
| C. reinhardtii | SpCas9 | S. pyogenes | Yes | NGG | |
| C. reinhardtii | Dead SpCas9(dCas9) | S. pyogenes | Maize codon–optimized | NGG | |
| P. tricornutum | SpCas9 | S. pyogenes | Yes | NGG | |
| N. oceanica IMET1 | SpCas9 | S. pyogenes | Yes | NGG | |
| N. gaditana | SpCas9 | S. pyogenes | Yes | NGG | |
| N. salina | SpCas9 | S. pyogenes | Chlamydomonas-codon optimized | NGG | |
| N. oceanica | dCas9 | S. pyogenes | Yes | NGG | |
| T. pseudonana | SpCas9 | S. pyogenes | Human-codon optimized | NGG | |
| T. pseudonana | SpCas9 | S. pyogenes | Yes | NGG | |
| C. vulgaris | SpCas9 | S. pyogenes | Yes | NGG | |
| C. vulgaris | SpCas9 | S. pyogenes | Maize-codon optimized | NGG | |
| C. sorokiniana | dCas9 | S. pyogenes | Maize-codon optimized | NGG |
| Features | Microalgal strains | Host | References | |
|---|---|---|---|---|
| Cas9 | pCaMV 35S/tNOS | C. reinhardtii | Cauliflower mosaic virus (CaMV) | |
| C. vulgaris | ||||
| C. sorokiniana | ||||
| pHSP70A-RBCS2/RBCS2 3’UTR | C. reinhardtii | C. reinhardtii | ||
| pPsaD/tPsaD | C. reinhardtii | C. reinhardtii | ||
| pLHCF2/tLHCF1 | P. tricornutum | P. tricornutum | ||
| pVCP /tATUB | N. oceanica IMET1 | N. oceanica IMET1 | ||
| pRPL24/ tFRD | N. gaditana | N. gaditana | ||
| pRibi/tldsp | N. oceanica | N. oceanica | ||
| pTpFCP/tNAT | T. pseudonana | T. pseudonana | ||
| pNR (nitrate reductase promoter)/tNR (nitrate reductase terminator) | T. pseudonana | T. pseudonana | ||
| pFcpB/tFcpA | P. tricornutum | P. tricornutum | ||
| sgRNA | pU6/T6 | C. reinhardtii | Arabidopsis | |
| C. reinhardtii | C. reinhardtii | |||
| P. tricornutum | P. tricornutum | |||
| T. pseudonana | T. pseudonana | |||
| C. vulgaris | Arabidopsis | |||
| C. sorokiniana | Arabidopsis | |||
| pATPase/tfd | N. oceanica IMET1 | N. oceanica IMET1 | ||
| pPsaD/tPsaD | C. reinhardtii | C. reinhardtii | ||
| pRibi/tcs | N. oceanica | N. oceanica | ||
| Donor DNA | pβ-tub/trbcS2 (for HygR) | C. reinhardtii | C. reinhardtii | |
| pVCP/tVCP (for Sh bleR) | N. oceanica IMET1 | N. oceanica IMET1 | ||
| pβ-tub/tCOP1 (for aphVII) | C. reinhardtii | C. reinhardtii | ||
| pHSP70A/RBCS2 3’UTR (for bacterial phytase gene) | C. reinhardtii | C. reinhardtii | ||
| pTUB2/tCOP21 (for aphVIII) | C. reinhardtii | C. reinhardtii | ||
| Selection marker | pCaMV 35S/tNOS (for HygR) | C. reinhardtii | CaMV | |
| pFCP/pFCP (for Sh bleR) | P. tricornutum | P. tricornutum | ||
| pHSP70-RBCS2/RBCS2-3UTR (for aphVIII) | C. reinhardtii | C. reinhardtii | ||
| pPsaD/tPsaD (for Sh bleR) | C. reinhardtii | C. reinhardtii | ||
| pTCT/ tEIF3 (for blasticidin deaminase) | N. gaditana | N. gaditana | ||
| pEIF3/tFRD (for HygR) | N. gaditana | N. gaditana | ||
| pUEP/tUEP (for Sh bleR) | N. salina | N. salina |
Mutagenesis efficiency (%) was determined by calculating the proportion of mutants confirmed through genotyping methods, such as Sanger sequencing or polymerase chain reaction (PCR), against the total number of mutants subjected to genotyping. Targeted mutagenesis efficiency was defined by the number of mutants confirmed by genotyping in relation to the initial size of the mutant pool.
Table 1.
Table 2.
Table 3.
Table 4.
TOP
 MSK
MSK
 ePub Link
ePub Link Cite this Article
Cite this Article





